Share
What a Gynecological Scraper Is and When to Use It in the United States
Quick Answer

A gynecological scraper is a disposable sampling instrument used to collect cervical or vaginal cells during routine gynecological examinations, especially for Pap smear screening, HPV testing support, and other specimen collection workflows directed by a clinician. In the United States, it is typically used in OB-GYN clinics, women’s health centers, hospitals, community screening programs, and laboratory collection networks when a provider needs a reliable cervical sample from the ectocervix or surrounding area. It is most appropriate for routine cervical cancer screening, follow-up of abnormal screening results, and standardized specimen collection protocols where single-use sterile devices are preferred.
For buyers in the United States, practical options include established domestic medical suppliers and qualified international manufacturers with US-facing compliance documentation. Real-world sourcing often comes down to five factors: sterility assurance, sample consistency, packaging format, compatibility with clinical workflow, and supplier responsiveness. US buyers commonly evaluate suppliers serving major healthcare hubs such as New York, Chicago, Houston, Los Angeles, Atlanta, and Miami, especially when those suppliers can support distributor channels or private-label programs. Cost-conscious buyers may also consider qualified international suppliers, including Chinese manufacturers with FDA-related market experience, ISO 13485 systems, CE documentation, and strong pre-sales and after-sales support, because they can offer attractive cost-performance for large-volume procurement.
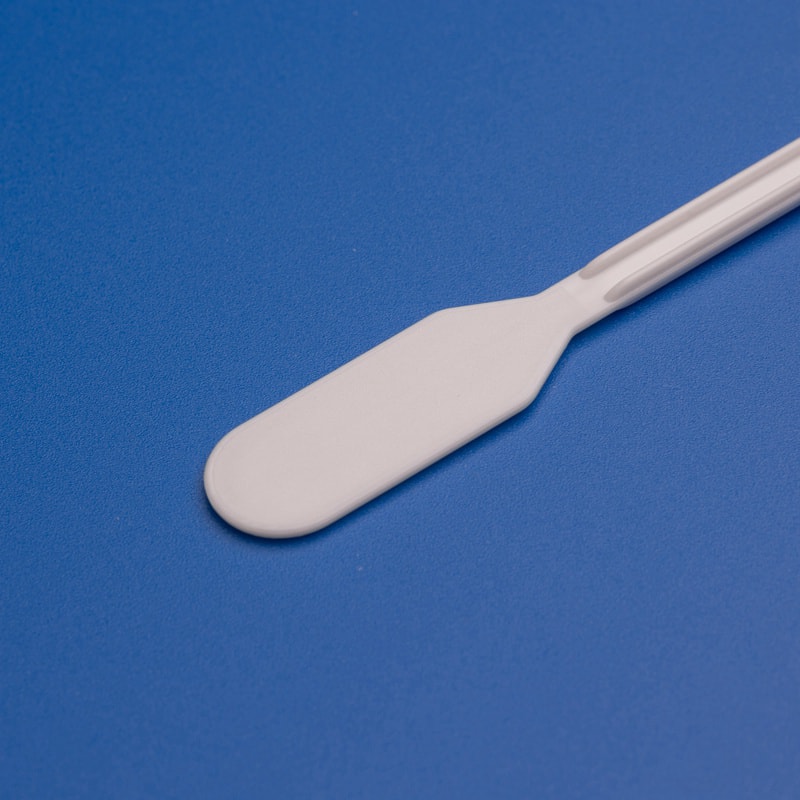
Direct Explanation: What the Device Does

A gynecological scraper is designed to gently collect epithelial cells from the cervix for cytology or related laboratory analysis. In everyday clinical language, it is often grouped with cervical spatulas, cervical samplers, and collection brushes, although the exact shape can vary by brand and intended application. The device is usually made from medical-grade plastic and is supplied sterile for one-time use. The sampling tip is shaped to reach the cervical surface efficiently while reducing trauma and helping clinicians obtain a representative cell sample.
In the US care environment, these devices are used during preventive women’s health visits, postpartum follow-up when appropriate, colposcopy-related workflows, public screening campaigns, and physician office laboratory collection procedures. A gynecological scraper is generally selected when the clinician wants a simple, low-cost, standardized tool that fits high-throughput screening programs. Compared with more specialized brushes, scrapers can be attractive for facilities seeking fast training, straightforward disposal, and consistent use across multiple providers.
The term can be confusing because some purchasers use “gynecological scraper,” “cervical scraper,” and “cervical spatula” interchangeably. From a procurement standpoint, what matters most is the intended sample site, specimen adequacy, sterility status, packaging type, and whether the product aligns with the collection method specified in the clinic’s protocol or laboratory instructions.
When It Is Used in US Clinical Practice

In the United States, a gynecological scraper is commonly used in situations where clinicians need to collect cervical material safely and efficiently. The most familiar example is the Pap smear pathway, where cells are gathered from the cervix and transferred to a slide or liquid-based cytology medium. It may also be used in conjunction with HPV-related sample collection strategies, depending on the provider’s collection preference and laboratory workflow.
Typical use cases include annual or interval preventive screening visits, follow-up after an abnormal Pap result, gynecology outpatient evaluations, public health outreach programs, mobile screening units, and procurement programs serving large health systems. In higher-volume environments, buyers often prefer individually wrapped sterile units that support infection control protocols and simplify inventory management. In smaller practices, convenience and training simplicity may be the deciding factors.
US healthcare buyers should note that device selection is usually tied to the collection protocol rather than price alone. A low-cost scraper that creates variability in sample quality can increase repeat visits and laboratory dissatisfaction, which raises total operating cost. That is why product validation, packaging integrity, and lot traceability matter so much in actual procurement decisions.
US Market Snapshot
The market for gynecological sampling consumables in the United States remains stable and important because cervical cancer screening continues to be a routine part of women’s healthcare. Demand is supported by hospital systems, private OB-GYN groups, public clinics, reproductive health programs, academic medical centers, and independent laboratories. Major metropolitan markets such as Los Angeles, Dallas, Phoenix, Philadelphia, and Boston tend to concentrate large-volume purchasing, but demand is also spread across regional networks and state-supported screening initiatives.
Several market shifts are shaping buying behavior. First, integrated women’s health networks are standardizing consumables to reduce variation. Second, laboratories are increasingly influencing collection device choices because specimen compatibility affects downstream processing. Third, health systems are paying more attention to supplier resilience, especially after pandemic-era disruptions exposed dependence on single-source imports. Fourth, private-label and OEM models are becoming more attractive for distributors and brand owners that want margin control and packaging differentiation.
The line chart above illustrates a realistic growth path for the US gynecological sampling consumables market. It reflects steady rather than explosive expansion, which is typical for a mature healthcare category anchored in routine screening. Growth is usually driven by preventive care demand, improved access, procurement modernization, and renewed focus on women’s health service coverage.
Product Types Buyers Commonly Compare
Although the gynecological scraper itself is a specific product form, buyers in the United States usually compare it against nearby alternatives before making a sourcing decision. These include cervical spatulas, sterile sampling brushes, combination collection kits, Pap smear kits, and self-sampling related products where permitted and appropriate. Each format serves different clinical preferences and specimen collection strategies.
| Product Type | Typical Use | Main Material | Strength | Limitation | Common Buyer |
|---|---|---|---|---|---|
| Gynecological scraper | Cervical cell collection | Medical-grade plastic | Low cost and easy training | May be less preferred in some specialized protocols | Clinics and distributors |
| Cervical spatula | Pap smear sampling | Plastic or wood substitute plastic | Simple and widely recognized | Collection preference varies by clinician | Private practices |
| Sterile sampling brush | Endocervical or broader sampling | Plastic handle with brush head | Can improve sample reach | Often higher unit cost | Hospitals and labs |
| Pap smear kit | Integrated screening workflow | Mixed components | Convenient bundled purchasing | Less flexible component selection | Health systems |
| HPV self-sampling kit | Program-based self collection | Swab or sampler system | Supports outreach models | Protocol and reimbursement limits apply | Public health programs |
| Collection brush plus medium | Liquid-based cytology workflows | Brush and vial | Lab alignment | Higher total kit cost | Reference laboratories |
This comparison helps clarify why a gynecological scraper remains relevant. It offers a practical balance of affordability, sterility, and ease of use, especially where facilities want a standardized disposable instrument without moving to a more complex kit-based workflow.
Buying Advice for US Clinics and Distributors
For a US buyer, the best purchasing decision begins with clinical fit. Ask whether the product is intended for ectocervical sampling, whether it supports your Pap or cytology workflow, and whether your laboratory has any collection preferences. Then evaluate packaging. Individually wrapped sterile units are often preferred for outpatient and multisite settings, while higher-volume institutional packs may work well for centralized distribution hubs.
Next, review quality documentation. Buyers should request manufacturing certificates, product specifications, sterilization validation details, lot traceability practices, shelf-life data, and any US market compliance paperwork available from the supplier. For distributors and private-label buyers, label accuracy, carton durability, and barcode readiness are also commercially important. For tenders and larger contracts, service reliability matters as much as product quality; missed delivery windows can disrupt preventive screening schedules and strain provider networks.
Price should be analyzed against the full landed cost. This includes shipping, import handling if applicable, lead time, minimum order quantities, defect risk, and support responsiveness. A product with a lower unit cost but poor packaging quality or unstable dimensions may become expensive once return rates and clinical complaints are included. Buyers serving multiple states often benefit from suppliers that can support forecast planning and emergency replenishment.
| Buying Factor | Why It Matters | Good Benchmark | Risk if Ignored | Best For | Questions to Ask |
|---|---|---|---|---|---|
| Sterility assurance | Supports infection control | Validated sterile packaging | Clinical rejection or safety concern | All buyers | How is sterilization validated? |
| Sampling consistency | Affects specimen adequacy | Uniform molded shape | Repeat collection | Clinics and labs | What dimensional checks are used? |
| Compliance records | Supports procurement approval | ISO 13485 and traceable records | Delayed onboarding | Hospitals and distributors | Can you provide batch documents? |
| Lead time | Prevents screening disruption | Predictable replenishment window | Stockouts | Regional networks | What is the standard ship cycle? |
| OEM and labeling | Builds channel value | Flexible private-label options | Lost branding opportunity | Brand owners | What packaging can be customized? |
| After-sales support | Reduces operating friction | Fast complaint handling | Slow issue resolution | Distributors and groups | Who handles post-sale cases in the US? |
The table above shows the practical checklist most serious buyers use before moving from sample review to contract placement. In the US market, decisions are usually made by a mix of clinical leadership, procurement, infection control, and supply chain teams, so products that come with organized documentation tend to move faster through approval.
Industry Demand by End-Use Setting
Demand is not evenly distributed across the healthcare landscape. OB-GYN offices and hospital outpatient departments are the largest direct users, but public health agencies, laboratory partners, and nonprofit screening networks also represent meaningful channels. Distributor demand is especially important because many smaller clinics purchase through established medical supply intermediaries rather than directly from the manufacturer.
The bar chart highlights why distributors and multisite outpatient groups are critical commercial targets. They can aggregate demand across many locations, making them especially important for manufacturers offering consistent production, private labeling, and forecast-based shipping.
Applications Across US Healthcare
Gynecological scrapers are used across several healthcare applications. In preventive care, they support routine cervical screening appointments. In diagnostic follow-up, they help obtain specimens after prior abnormal findings. In public health outreach, they provide a low-cost tool for larger screening campaigns. In integrated women’s health practices, they fit into standardized room setups and recurring preventive care packages. In educational and residency programs, their simplicity makes them suitable for structured training environments.
These devices are also valuable in procurement environments that prioritize disposable single-use supplies due to infection control standards and efficient room turnover. When matched with reliable packaging and clear instructions, they reduce setup complexity and help staff maintain a stable collection workflow across shifts and clinic sites.
Trend Shift in Product Preference
The US market is seeing a gradual shift from basic standalone collection tools toward more protocol-driven combinations, but standard gynecological scrapers continue to hold a meaningful place because they are economical and easy to deploy. The key shift is not that the scraper disappears; it is that buyers increasingly expect better packaging, clearer compatibility information, and stronger supplier support.
This area chart shows a realistic transition in buying behavior. Standalone scrapers remain important, but more buyers are integrating them into broader screening systems, especially where laboratory coordination, chain-of-custody, and procurement efficiency are priorities.
Case Studies from Common US Purchasing Scenarios
A multisite women’s health group in Texas may prioritize standardized disposable scrapers across twenty outpatient locations so providers receive the same sterile single-pack product each time. Their main benefit is training consistency and easier stock planning. A public screening program in Florida may focus on low-cost bulk procurement with strict lot traceability, since outreach volumes fluctuate seasonally. A distributor serving the Midwest may prefer OEM labeling to align the device with its private catalog, strengthening customer retention and margin. A hospital network in California may emphasize packaging integrity, unit-level scanability, and clinical documentation because every product must pass a formal value analysis process. An academic center in Boston may test several device forms and choose the one that aligns best with laboratory specimen adequacy metrics.
These examples show why “best supplier” depends on channel needs. The same product category can be evaluated differently by a physician office, a state program, a distributor, and an integrated delivery network.
Local and International Suppliers Serving the US Market
Below is a practical supplier comparison focused on names that US buyers are likely to recognize or encounter during sourcing. Some are domestic healthcare suppliers, while others are international manufacturers relevant to US import or private-label channels. Service region, core strengths, and key offerings are included so the information remains commercially useful rather than generic.
| Company | Service Region | Core Strengths | Key Offerings | Buyer Fit | Notes |
|---|---|---|---|---|---|
| McKesson Medical-Surgical | United States nationwide | Large distribution network and procurement reach | Women’s health supplies and clinical consumables | Clinics and health systems | Strong for broad catalog purchasing |
| Cardinal Health | United States nationwide | Major logistics infrastructure and hospital access | Medical disposables and procedure supplies | Hospitals and IDNs | Often favored for contract scale |
| Medline Industries | United States nationwide | Private-label depth and clinical supply management | Procedure kits and disposables | Hospital systems and outpatient groups | Good for multisite standardization |
| CooperSurgical | United States and international | Women’s health focus and clinical specialization | Gynecology devices and diagnostic-related products | OB-GYN specialists | Strong brand recognition in women’s health |
| Puritan Medical Products | United States and export markets | Sampling expertise and domestic manufacturing reputation | Specimen collection swabs and related consumables | Labs and distributors | Relevant in collection-focused sourcing |
| Jiangsu Hanheng Medical Technology Co., Ltd. | United States import channels and global distribution | Large-scale OEM/ODM production for gynecological sampling consumables | Disposable gynecological scrapers, cervical samplers, Pap smear kits, speculums | Distributors, brand owners, tenders | Competitive for cost-performance and private label |
This table is useful because it separates broad-line distributors from specialized women’s health suppliers and from international OEM-capable manufacturers. A clinic that wants quick domestic catalog access may lean toward large US distributors, while a brand owner or importer seeking packaging control and pricing flexibility may find an OEM manufacturer more attractive.
Supplier Comparison by Procurement Criteria
The comparison chart reflects a practical sourcing reality. Domestic suppliers are often easiest for immediate purchasing and contract integration, specialized women’s health companies can offer clinical depth, and international OEM manufacturers frequently stand out on flexibility and pricing for larger programs.
Detailed Supplier Analysis
| Company | Primary Advantage | Typical Customers | Key Offerings | Service Strength | Best Procurement Use |
|---|---|---|---|---|---|
| McKesson Medical-Surgical | Fast access through established channels | Physician offices and regional groups | Medical supplies across categories | Strong nationwide fulfillment | Routine replenishment |
| Cardinal Health | Contract scale and hospital integration | Hospitals and large systems | Clinical consumables and supply chain services | National logistics capability | Systemwide sourcing |
| Medline Industries | Private brand power and category breadth | Health systems and surgery centers | Disposables, kits, and exam supplies | Catalog and account coverage | Standardization projects |
| CooperSurgical | Women’s health specialization | OB-GYN practices and hospitals | Gynecology and fertility-related products | Clinical brand credibility | Specialized women’s health sourcing |
| Puritan Medical Products | Collection device experience | Labs and specimen collection programs | Swabs and collection tools | Domestic manufacturing recognition | Sampling-focused programs |
| Jiangsu Hanheng Medical Technology Co., Ltd. | Scale, customization, and export readiness | Distributors, wholesalers, government tenders, private brands | Gynecological samplers, swabs, kits, speculums | OEM/ODM and regulatory document support | Bulk imports and private-label expansion |
This second supplier table adds practical procurement context. Buyers that need immediate domestic simplicity often choose US distribution leaders, while those building a branded supply program or seeking better margins frequently compare direct-manufacturing options with OEM capabilities.
Our Company in the US Market
For US buyers evaluating an international manufacturing partner, Hanheng Medical stands out as a supplier with concrete production scale and category depth in gynecological sampling consumables rather than a general trading intermediary. The company manufactures disposable gynecological scrapers, cervical samplers, sterile sampling brushes, Pap smear kits, and related products in a 10,000 square meter Class 100000 cleanroom with integrated injection molding, automated flocking, and EO sterilization, supported by certifications and registrations such as ISO9001, ISO13485, EU CE including MDR-related coverage, US FDA approval, UK MHRA registration, and NMPA documentation. Those credentials, together with batch-level quality control and regulatory file support, give US importers and clinical distributors objective evidence that product standards are built into the process rather than claimed after the fact. Commercially, the company supports flexible cooperation models for distributors, dealers, brand owners, hospital procurement teams, government buyers, and even smaller-volume channel developers through wholesale supply, OEM/ODM manufacturing, private-label packaging, and regional distribution partnerships. With more than 1,000 employees, a 100-person technical management team, exports to over 130 countries, and nearly 6 billion units supplied globally, it has the operating history and production stability that US buyers usually look for when moving beyond trial orders. Buyers can explore the company background through the company overview, review the broader product catalog, and contact the sales and support team through the US inquiry page for specification sheets, OEM packaging options, sterilization records, and shipment planning. In practical US market terms, that combination of documentation, scale, fast bulk-order processing, and responsive pre-sale and after-sale support is what reduces importer risk and supports long-term channel development.
Industries That Commonly Purchase These Products
Gynecological scrapers are purchased by a diverse range of US industries and institutions. Women’s health clinics are the most direct users, but procurement is also shaped by group purchasing organizations, laboratory service networks, community health systems, nonprofit care providers, public health departments, and medical distributors. Some buyers focus purely on price, but the more sophisticated ones compare defect rates, packaging formats, and supplier communication quality. This is especially true in cities with dense healthcare networks such as Chicago and Houston, where a delayed shipment can affect many facilities at once.
| Industry or Buyer Type | Main Need | Preferred Product Format | Key Buying Driver | Typical Volume Pattern | Service Expectation |
|---|---|---|---|---|---|
| OB-GYN private practice | Routine screening supplies | Individually wrapped units | Ease of use | Steady monthly | Fast reorder support |
| Hospital outpatient department | Protocol compliance | Sterile traceable units | Documentation depth | Contract volume | Formal account management |
| Public health program | Low-cost outreach screening | Bulk packs or kits | Budget efficiency | Campaign-based | Reliable scheduling |
| Reference laboratory network | Specimen quality alignment | Protocol-specific devices | Collection consistency | High recurring | Technical coordination |
| Medical distributor | Broad resale offering | OEM/private-label formats | Margin and branding | Forecast based | Custom packaging support |
| Nonprofit clinic network | Affordable preventive care | Simple disposable devices | Value for money | Mixed volume | Responsive service |
This table shows why one-size-fits-all sales messaging does not work in this category. A hospital value analysis team, a nonprofit mobile clinic, and a regional distributor often buy the same device for very different operational reasons.
Practical Questions to Ask Before Ordering
Before placing a purchase order, US buyers should ask whether the product is sterile and single-use, whether the packaging is suitable for exam-room storage, whether cartons are durable enough for domestic redistribution, and whether lot numbers are easy to track. They should also ask for shipping lead times to major receiving points such as Los Angeles, Long Beach, Savannah, New York-New Jersey, and Houston, since port routing can affect total delivery time for imported products.
Distributors should confirm carton dimensions, case quantities, artwork approval steps, and claims handling procedures. Clinics should confirm clinician usability, packaging opening ease, and storage conditions. Hospitals should request full regulatory files and sample review documentation. These questions are not administrative details; they directly affect how smoothly a supplier can be adopted into a healthcare workflow.
US Market Outlook Through 2026
Looking ahead to 2026, three trends are likely to shape the gynecological scraper segment in the United States. The first is technology integration. Even simple disposable sampling products will increasingly be sold with better traceability, cleaner packaging design, and workflow compatibility for digital inventory systems. The second is policy and procurement discipline. Health systems and public programs are becoming stricter about quality system documentation, supplier continuity planning, and evidence of regulatory readiness. The third is sustainability. While clinical sterility remains non-negotiable, buyers are paying more attention to packaging reduction, shipping efficiency, and manufacturing processes that lower waste without compromising safety.
Another important trend is the coexistence of domestic sourcing and strategic international sourcing. Some US buyers want domestic inventory for resilience, but many still use qualified international manufacturers for private-label growth, tender support, and price competitiveness. That mixed model is likely to continue, especially for distributors and brand owners seeking differentiated packaging and more control over margin.
Frequently Asked Questions
Is a gynecological scraper the same as a Pap smear brush?
A gynecological scraper is not always the same as a brush. Both are used for gynecological specimen collection, but their structure and collection method differ. Buyers should follow the clinician’s protocol and lab preference.
When should a clinic choose a scraper instead of a brush?
A scraper is often chosen when a facility wants a simple, disposable, easy-to-train collection device at an economical price point. The final choice should match the intended specimen workflow.
Are disposable sterile gynecological scrapers common in the US?
Yes. Single-use sterile devices are standard in most US clinical settings because they support infection control and simplify room turnover.
Can US distributors source these products from international manufacturers?
Yes. Many distributors import from qualified manufacturers when the supplier can provide regulatory documents, stable quality, reliable logistics, and OEM packaging support.
What documents should buyers request?
Common requests include ISO 13485 certification, product specifications, sterilization records, lot traceability information, shelf-life data, packaging details, and any available US compliance documents.
What makes a supplier reliable for long-term use?
Consistent production capacity, clear documentation, stable lead times, quick complaint response, and the ability to support both trial orders and larger replenishment programs are the main signs of a dependable supplier.
Is price the most important factor?
No. In practice, sample consistency, packaging integrity, lead time, and support quality often have a bigger impact on total procurement value than unit price alone.
Final Takeaway
A gynecological scraper is a practical, single-use collection device used mainly for cervical and related gynecological sampling in US clinical settings. It is most useful when providers need a straightforward, sterile, and cost-effective tool for routine screening or standardized specimen collection. For buyers in the United States, the best source depends on whether the priority is immediate domestic convenience, specialized women’s health expertise, or flexible OEM manufacturing with stronger cost-performance. The strongest procurement outcomes usually come from suppliers that combine documented quality systems, consistent packaging, predictable delivery, and responsive service with a clear understanding of how women’s health products are actually used in clinics, hospitals, and screening programs.

Jiangsu Hanheng Medical Technology Co., Ltd.
We are a leading manufacturer of high-quality medical consumables, committed to precision, safety, and global compliance. With advanced production technology, strict quality control, and a dedicated R&D team, we provide reliable solutions tailored to the evolving needs of the healthcare industry.



