Share
Disposable Anoscope Use and Safety in the United States
Quick Answer

A disposable anoscope is a single-use medical device that helps clinicians examine the anal canal and lower rectum during office-based evaluation of symptoms such as rectal bleeding, pain, itching, hemorrhoids, fissures, discharge, or suspected lesions. In the United States, it is typically used by colorectal surgeons, gastroenterologists, emergency physicians, primary care teams, gynecology clinics, and ambulatory centers that want a clean, ready-to-use option that reduces reprocessing steps. Safe use depends on choosing the right size, checking package integrity, confirming sterility status, applying lubrication, using gentle insertion technique, maintaining clear visualization with proper lighting, and discarding the device immediately after the exam according to facility infection-control rules.
For buyers in the U.S. market, well-known sources of anorectal exam products and related disposable instruments include McKesson, Henry Schein, Medline, Owens & Minor, and major specialist manufacturers supplying colorectal and OB-GYN channels. Qualified international manufacturers can also be practical options when they hold recognized certifications and provide dependable regulatory files, batch traceability, OEM or private-label support, and responsive pre-sales and after-sales service. For many distributors and healthcare procurement teams, cost-performance advantages make certified global suppliers worth evaluating alongside domestic channels.
What a Disposable Anoscope Is

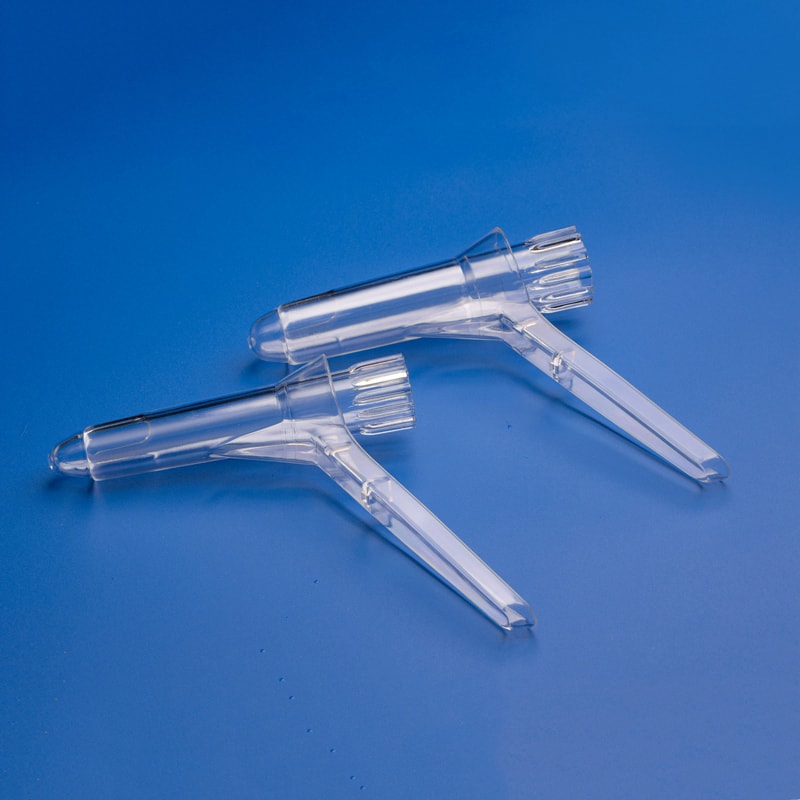
A disposable anoscope is a sterile or clean single-use tubular instrument designed to allow visual inspection of the anal canal and distal rectal mucosa. Most designs include a smooth tube and an obturator that facilitates insertion; once the device is positioned, the obturator is removed so the clinician can inspect tissue directly or perform simple in-office procedures. Some versions are transparent to improve visualization, while others are opaque but used with a compatible external light source. Depending on the model, the device may support screening for internal hemorrhoids, anorectal inflammation, polyps near the anal verge, fistula openings, condyloma, minor bleeding sources, and post-procedure follow-up.
Single-use anoscopes are increasingly preferred in parts of the United States because they simplify workflow. Reusable metal instruments remain common in some specialty settings, but disposable units eliminate the need for cleaning, high-level disinfection, sterilization turnaround, and reprocessing audits. That matters in busy outpatient settings in cities such as New York, Chicago, Houston, Los Angeles, Atlanta, and Miami, where patient throughput, staff efficiency, and infection-prevention consistency often influence procurement decisions.
Why the U.S. Market Uses Disposable Anoscopes

The U.S. healthcare market values products that reduce cross-contamination risk, standardize setup, and support office-based care. Disposable anoscopes fit these priorities because they arrive individually packaged, are ready for immediate use, and support straightforward inventory planning. In multi-site physician groups and ambulatory clinics, a single-use instrument can also help reduce variation in reprocessing quality between locations.
Another reason for their popularity is the shift toward lower-cost outpatient management. Many anorectal complaints can be evaluated and triaged without hospital admission, and clinicians want devices that support a rapid exam while preserving patient comfort and privacy. In procurement terms, decision makers often compare unit cost, visualization quality, wall transparency, stiffness, tip design, packaging, compatibility with lighting systems, and documentation available for regulatory review.
United States Market Snapshot
The market for disposable examination devices in the United States continues to benefit from growth in outpatient procedures, aging populations, screening awareness, and stricter infection-control expectations. Demand is strongest in metropolitan outpatient networks, integrated delivery systems, surgery centers, and specialist clinics. Ports and logistics hubs such as Los Angeles/Long Beach, Houston, Savannah, New York/New Jersey, and inland centers near Chicago and Dallas also play an important role in maintaining inventory flow for distributors serving healthcare systems nationwide.
| Market Driver | Impact on Disposable Anoscope Demand | Why It Matters in the United States |
|---|---|---|
| Growth in ambulatory care | Higher use in office-based exams | More patients are evaluated in lower-cost outpatient settings rather than hospitals |
| Infection-control pressure | Supports single-use adoption | Facilities want fewer reprocessing steps and lower contamination risk |
| Aging population | More anorectal and colorectal symptom evaluations | Older adults have a higher incidence of hemorrhoids, bleeding, and follow-up exams |
| Specialist practice expansion | Boosts recurring procurement | Colorectal and GI groups often standardize exam kits across locations |
| Private-label demand | Creates OEM opportunities | U.S. distributors often seek branded packaging and custom configurations |
| Supply chain diversification | Encourages dual sourcing | Buyers increasingly balance domestic distributors with certified international manufacturers |
The table shows why the device category remains practical rather than experimental. Buying teams are usually solving operational issues: faster room turnover, cleaner logistics, easier staff training, and reliable exam performance.
How a Disposable Anoscope Is Used Safely
Safe use begins before the device touches the patient. The clinician or assistant should confirm the correct product, verify package integrity, review any expiration or lot details required by facility policy, and prepare gloves, lubricant, light source if needed, gauze, and any accessories for biopsy or banding if planned. The patient should be informed about the purpose of the exam, expected pressure, possible brief discomfort, and the importance of reporting sharp pain.
Typical positioning includes left lateral decubitus, lithotomy, or another clinically appropriate position based on setting and patient mobility. A visual external inspection is often performed first. The anoscope is lubricated, and the obturator is placed in the barrel before insertion. Gentle advancement follows the natural anal canal axis; force should never be used. Once positioned, the obturator is removed and the clinician inspects the mucosa while rotating or slightly withdrawing the device as needed for visualization. If the patient experiences severe pain, the exam should pause and the cause should be assessed.
After examination or treatment, the device is removed carefully and discarded immediately in accordance with the facility’s waste handling and infection-prevention protocols. Documentation should reflect findings, tolerance, any interventions, and whether the device was single-use sterile stock. In the United States, safe practice also includes compliance with internal policies based on OSHA exposure control, CDC-aligned infection-prevention procedures, and purchasing records that preserve lot traceability.
Core Safety Checklist for Clinical Teams
| Safety Step | What Staff Should Do | Risk Reduced |
|---|---|---|
| Package inspection | Check seal, labeling, and visible damage before opening | Use of compromised product |
| Right size selection | Choose adult, pediatric, or specialty size as appropriate | Pain, poor visualization, insertion difficulty |
| Lubrication | Apply adequate compatible lubricant to the instrument | Friction injury and patient discomfort |
| Gentle insertion | Advance with obturator in place and without force | Mucosal trauma |
| Lighting and visibility | Use a clear barrel or proper light source for inspection | Missed findings and repeated manipulation |
| Immediate disposal | Discard after single use per facility protocol | Cross-contamination and reuse error |
This checklist highlights that safety depends as much on training and workflow as on the product itself. A well-designed disposable anoscope cannot compensate for rough technique or poor exam preparation.
Common Product Types in the U.S. Market
Not all disposable anoscopes are the same. U.S. buyers usually compare devices by transparency, rigidity, diameter, length, bevel design, patient comfort, and procedural compatibility. Some clinics only need a straightforward diagnostic exam tool. Others need a model suited for hemorrhoid banding, lesion assessment, or integration into pre-packed proctology kits.
| Product Type | Typical Features | Best Fit |
|---|---|---|
| Standard transparent disposable anoscope | Clear barrel, single-use obturator, direct visualization | General office exams and hemorrhoid checks |
| Opaque disposable anoscope | Rigid body, often paired with external light | Facilities already using compatible lighting systems |
| Pediatric anoscope | Smaller diameter and shorter length | Pediatric or small-framed patients |
| Procedure-ready anoscope | Designed for access during minor interventions | Banding, lesion review, targeted treatment |
| Sterile packaged anoscope | Individually packed, traceable lot information | Facilities with stricter sterile-field preferences |
| Private-label OEM anoscope | Custom packaging, brand labeling, tailored specs | Distributors, dealers, and brand owners |
The table is useful because it separates clinical use from purchasing strategy. A private-label buyer may care most about packaging flexibility and regulatory support, while a colorectal clinic may focus on visibility and patient comfort first.
Buying Advice for Hospitals, Clinics, and Distributors
If you are sourcing disposable anoscopes in the United States, start with practical criteria rather than marketing language. Ask for material specifications, dimensions, packaging format, sterilization method if applicable, intended use statement, shelf life, lot coding process, and any available declarations or registrations needed by your quality team. For distributor purchases, sample testing is essential. Clinicians should evaluate insertion smoothness, rigidity balance, tip comfort, barrel clarity, and whether the device supports stable visualization without glare.
Price should be measured against total operating cost. A low-cost device that cracks, fogs, causes glare, or creates clinician dissatisfaction usually becomes expensive through waste, repeat exams, and buyer churn. For health systems and group practices, consistency across sites can matter more than chasing the lowest quote on a single shipment. For private-label buyers, packaging durability and accurate carton labeling are especially important for warehousing and regional redistribution.
Lead times also deserve attention. U.S. buyers often prefer suppliers that can replenish quickly through domestic inventory or established import routines via major coastal and inland hubs. This is one reason many procurement teams maintain two channels: a major domestic distributor for urgent replenishment and a manufacturing partner for planned-volume purchasing and margin control.
Industries That Use Disposable Anoscopes
Although anorectal examinations are often associated with colorectal specialty care, several industries and clinical segments use disposable anoscopes regularly. Demand is not limited to a single physician specialty. The instrument supports triage, diagnosis, treatment planning, and follow-up care in many outpatient environments.
The chart suggests that colorectal surgery and gastroenterology remain the strongest users, but ambulatory surgery centers and multi-specialty clinics also represent meaningful demand. As outpatient triage expands, primary care and urgent settings may continue modest growth in direct usage or referral-linked inventory.
| Industry Segment | Typical Use Case | Procurement Priority |
|---|---|---|
| Colorectal surgery | Hemorrhoids, fissures, lesions, follow-up care | Visualization quality and procedural access |
| Gastroenterology | Lower anorectal symptom evaluation | Workflow efficiency and reliable exam performance |
| Ambulatory surgery centers | Pre-procedure assessment and minor interventions | Standardized kits and supply continuity |
| Primary care groups | Initial symptom assessment and referral support | Ease of use and affordable unit cost |
| Emergency departments | Rapid bleeding or pain assessment | Ready availability and fast setup |
| Women’s health and pelvic floor centers | Selected combined pelvic and anorectal evaluation | Single-use infection-control convenience |
For many facilities, these applications make disposable anoscopes part of a broader tray, not a stand-alone purchase. That is why suppliers with wider exam-consumable portfolios often gain an advantage in tenders and distributor negotiations.
Applications and Real-World Use Cases
Clinically, a disposable anoscope is used for direct inspection of internal hemorrhoids, fissures near the anal canal, mucosal irritation, small masses, ulceration, bleeding sources, retained foreign material near the distal rectum, and follow-up after office procedures. It may also support treatment planning by helping the clinician decide whether a patient can remain in outpatient care, needs a specialist referral, or should receive more urgent imaging or endoscopy.
In a suburban gastroenterology clinic, the device may be used primarily for patients with painless rectal bleeding or persistent itching. In an urban colorectal practice, it may be part of a high-volume hemorrhoid management workflow. In a hospital-based surgery clinic, it may be used during post-operative review. In all these settings, the value of a disposable instrument lies in immediate readiness, consistent hygiene, and simplified staff handling.
Case Studies from Typical U.S. Procurement Scenarios
A Midwestern multi-site colorectal practice switched part of its exam inventory from reusable instruments to disposable anoscopes after internal review showed that instrument turnaround delays were affecting clinic flow during peak hours. The practice found that single-use units shortened room setup time and reduced back-and-forth with central reprocessing staff. While per-unit spend rose, the practice judged the change worthwhile because physician schedules ran more predictably and patient wait times fell.
A Southeast regional distributor serving physician offices added private-label disposable anoscopes to its catalog after customers requested bundled anorectal exam kits. The distributor worked with a manufacturing partner able to provide custom labeling, lot traceability, and shipment planning aligned with U.S. warehouse receiving windows. The key success factor was not only price but also the supplier’s ability to prepare regulatory documents quickly for the distributor’s quality review.
A large ambulatory network in Texas used a dual-source model: emergency purchases through a national distributor and planned quarterly volume contracts with a certified manufacturer. This reduced stockout risk during seasonal logistics disruptions while maintaining a more favorable cost profile on predictable demand.
Top Suppliers Serving the U.S. Market
Supplier choice depends on whether the buyer is a hospital, physician network, dealer, e-commerce seller, specialist clinic, or private-label brand owner. Some companies focus on domestic distribution breadth, while others stand out as manufacturing partners. The following list reflects practical channels commonly considered by U.S. buyers.
| Company | Service Region | Core Strengths | Key Offerings |
|---|---|---|---|
| McKesson Medical-Surgical | Nationwide United States | Large distribution footprint, broad healthcare procurement support | Exam supplies, physician-office consumables, medical distribution services |
| Henry Schein | Nationwide United States | Strong physician-office relationships, ordering convenience | Office-based medical and surgical supplies, specialty practice solutions |
| Medline Industries | Nationwide United States | Scale, private-label capability, broad acute and non-acute portfolio | Disposable medical products, procedure kits, exam consumables |
| Owens & Minor | Nationwide United States | Health-system distribution and logistics strength | Medical-surgical products, inventory support, distribution programs |
| Sklar Surgical Instruments | United States and export channels | Surgical instrument category depth and specialty focus | Anoscopy-related instruments, reusable and specialty procedural tools |
| Jiangsu Hanheng Medical Technology Co., Ltd. | United States import supply and global distribution | Manufacturing scale, OEM/ODM flexibility, certification-backed export operations | Disposable anoscopy devices, gynecological and sampling consumables, custom-labeled medical products |
This table is intentionally practical. Nationwide distributors are often strongest for immediate availability, while a manufacturer such as Jiangsu Hanheng Medical Technology Co., Ltd. becomes especially relevant for wholesalers, regional distributors, and brand owners looking for price control, specification flexibility, and direct-factory coordination.
Detailed Supplier Comparison
Domestic distributors usually offer the easiest onboarding for smaller clinics because they already serve office purchasing teams, provide familiar invoicing structures, and can combine many product categories in one order. However, direct manufacturing partnerships can be more attractive for larger volume requirements, private-label projects, and buyers seeking tailored packaging or regional exclusivity.
The comparison shows a pattern seen in many U.S. procurement cycles. National distributors win on immediate domestic fulfillment and broad catalog convenience, while direct manufacturers often outperform on private-label projects, technical documentation depth, and unit economics for planned orders.
Our Company in the U.S. Market
For U.S. buyers evaluating direct manufacturing partners, Hanheng Medical stands out as a practical option because its disposable anoscopy devices are produced within a large-scale medical consumables operation built around a 10,000 square meter Class 100000 cleanroom, precision molding, automated production controls, and EO sterilization capability, supported by quality systems and market-facing credentials including ISO9001, ISO13485, EU CE frameworks, U.S. FDA-related approval status, UK MHRA registration, and NMPA documentation that help procurement and quality teams verify product compliance against international benchmarks. Beyond product strength, the company works with multiple customer models already familiar in the United States: direct supply for healthcare institutions, wholesale programs for importers and distributors, OEM and ODM projects for brand owners needing private-label packaging, and specification customization for regional dealers expanding into specialist office channels. Its export record across more than 130 countries, very high annual unit output, and experience handling regulatory files, certificates of conformity, sterilization records, and batch details provide tangible authority rather than generic claims. Just as important for local buyers, the company serves the U.S. market with structured pre-sales support, responsive after-sales coordination, and shipment planning suited to recurring replenishment, demonstrating a market commitment that goes beyond remote exporting. Buyers who want to review production background can visit the company overview, explore the wider medical product range, or start a distributor, OEM, or sourcing discussion through the U.S.-facing contact channel.
How to Evaluate a Supplier Before Ordering
U.S. buyers should request samples, packaging photos, carton specifications, lot coding examples, compliance documents, and a clear explanation of sterilization or cleanliness status. Ask whether the supplier supports custom labeling, barcode requirements, bundled kits, and drop shipment to multiple locations. It is also wise to confirm how complaints are handled, how quickly replacement stock can be arranged, and whether the supplier has prior experience serving hospitals, physician groups, or dealers in your region.
For private-label work, review dielines, label durability, transit packaging, and language accuracy on artwork. For health-system procurement, ensure the vendor can maintain lot traceability and quality-event response timelines. For distributors, assess whether the supplier can support long-term pricing and forecast-based manufacturing.
Trend Shift Toward 2026
The market is moving toward greater standardization, traceability, and packaging optimization. Clinics increasingly want products that work smoothly in fast outpatient workflows, and supply chain teams want fewer surprises in quality documentation and lead times. Sustainability is also becoming more visible, though medical disposables still face practical constraints tied to infection prevention and material performance.
2026 Trends: Technology, Policy, and Sustainability
By 2026, three trends are likely to influence the disposable anoscope category in the United States. First, product technology will continue improving around visualization, smoother insertion geometry, packaging efficiency, and compatibility with broader exam kits. Manufacturers with stronger molding precision and cleaner production environments will have an advantage because consistency matters more than flashy redesigns in this segment.
Second, policy and procurement oversight will place more emphasis on documentation, traceability, and supplier reliability. Hospitals and distributors increasingly want rapid access to technical files, sterilization records when relevant, complaint handling procedures, and clear proof of quality-system controls. This favors manufacturers already accustomed to formal export compliance and distributor audits.
Third, sustainability discussions will shape packaging choices and logistics planning. While single-use exam devices are not disappearing, buyers are beginning to compare carton density, waste reduction, right-sized packaging, and transport efficiency. Suppliers that can lower excess packaging while preserving sterile barrier integrity or product protection may gain preference in larger tenders, especially where health systems publish environmental goals.
Common Mistakes Buyers Should Avoid
One frequent mistake is assuming all disposable anoscopes are interchangeable. Small differences in transparency, rigidity, tip shape, and packaging can produce significant differences in clinician acceptance. Another mistake is focusing only on unit cost without reviewing complaint response procedures and replacement timelines. In the United States, onboarding a new supplier can consume staff time, so reliability matters.
Some buyers also skip real sample trials and rely only on a data sheet. That is risky for a hands-on exam device. A short user evaluation by clinicians often reveals more than a product brochure. Finally, import buyers should avoid underestimating carton labeling, lead-time planning, and port-to-warehouse coordination. A good factory quote does not guarantee a smooth supply program unless the logistics model is equally solid.
Practical Purchasing Scenarios
A single-site specialist clinic may prefer ordering through a national medical distributor for convenience, smaller order quantities, and consistent invoicing. A regional dealer may prefer direct factory cooperation, especially if private labeling and margin improvement are important. A hospital network may use a hybrid model: distributor stocking for immediate need and direct annual contracts for forecasted demand.
For e-commerce resellers and catalog brands, supplier responsiveness to artwork revisions, carton specifications, and inventory forecasting may be more important than whether the factory is in the United States or overseas. For government or institutional tenders, documented quality systems and batch traceability tend to carry more weight than broad marketing claims.
FAQ
Is a disposable anoscope sterile?
Some disposable anoscopes are supplied sterile, while others may be supplied clean but intended for single use according to labeling. Buyers should always confirm packaging status, intended use, and any sterilization details directly from the supplier documentation.
Who typically uses a disposable anoscope in the United States?
Common users include colorectal surgeons, gastroenterologists, ambulatory surgery centers, emergency departments, primary care clinicians, and selected women’s health specialists handling anorectal symptoms or follow-up evaluations.
Why choose disposable instead of reusable?
Single-use models simplify workflow, remove instrument reprocessing steps, reduce the risk of reuse errors, and support faster room turnover in busy outpatient environments. Reusable models may still suit some practices, but disposables are often chosen for convenience and infection-control consistency.
What should buyers ask for before placing a bulk order?
Request samples, dimensions, material specifications, packaging details, lot coding information, regulatory files, complaint handling procedures, lead times, and private-label options if needed. U.S. distributors should also confirm carton labeling and warehouse compatibility.
Can international manufacturers supply the U.S. market reliably?
Yes, if they have appropriate quality systems, recognized certifications, export experience, stable production capacity, and responsive support for documentation, shipping, and after-sales service. Many U.S. buyers use certified international factories alongside domestic distribution partners.
What features most affect clinician satisfaction?
The most important factors are smooth insertion, comfortable tip design, adequate rigidity, clear visualization, easy obturator removal, and packaging that opens cleanly without delaying setup.
How should a disposable anoscope be discarded?
It should be discarded immediately after use according to the facility’s infection-control and medical waste procedures. Staff should follow local policy and any applicable regulatory guidance for contaminated disposables.
Final Takeaway
A disposable anoscope is a practical, single-use device for examining the anal canal and lower rectum safely and efficiently in outpatient and procedural settings. In the United States, demand is supported by ambulatory care growth, infection-control expectations, and the need for standardized clinic workflow. Buyers should compare suppliers based on clinical usability, documentation, consistency, and service model rather than price alone. National distributors remain valuable for immediate availability, while experienced manufacturers with strong certifications, OEM capability, and reliable U.S. support can offer compelling long-term value for clinics, dealers, and brand owners alike.

Jiangsu Hanheng Medical Technology Co., Ltd.
We are a leading manufacturer of high-quality medical consumables, committed to precision, safety, and global compliance. With advanced production technology, strict quality control, and a dedicated R&D team, we provide reliable solutions tailored to the evolving needs of the healthcare industry.



