Share
What a Cervical Cytology Brush Is in the United States
Quick Answer

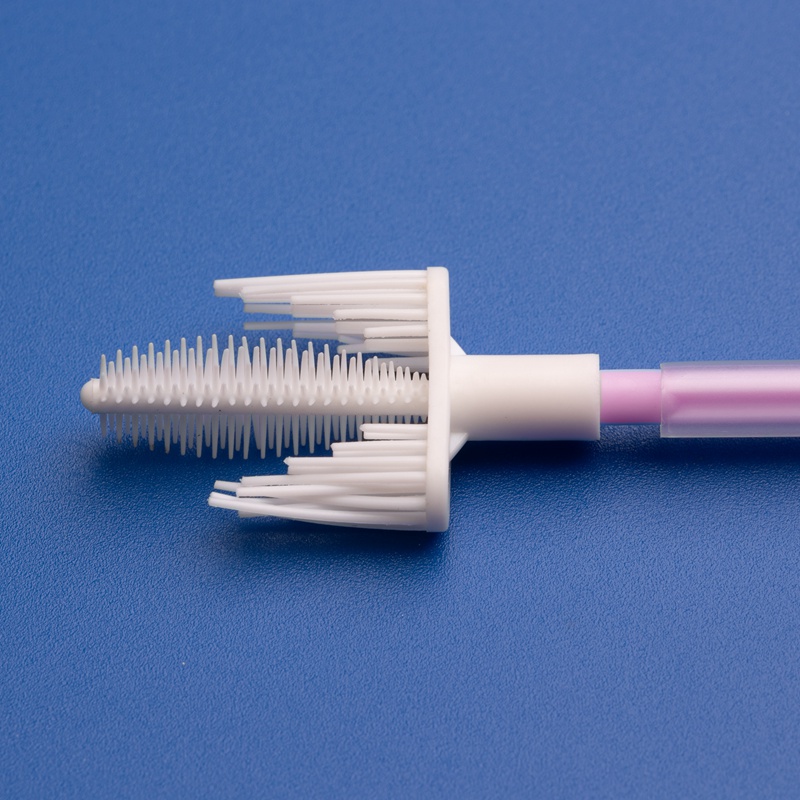
A cervical cytology brush is a single-use medical sampling device designed to collect cells from the cervix for cytology testing, most commonly for Pap tests and HPV-related screening. Clinics in the United States use it because the soft bristles are shaped to reach the cervical transformation zone, where abnormal cells are most likely to be found, while helping providers collect a consistent specimen with less trauma than older rigid tools.
In practical terms, U.S. clinics choose cervical cytology brushes when they want reliable sample adequacy, compatibility with liquid-based cytology systems, predictable workflow in OB-GYN offices, community health centers, hospital outpatient departments, and women’s health programs, and a disposable sterile device that supports infection-control protocols.
Common providers active in the U.S. market include Hologic, CooperSurgical, Medgyn Products, Puritan Medical Products, Cardinal Health, and McKesson private-label channels. Qualified international manufacturers can also be worth considering when they hold relevant certifications and provide strong technical documentation, consistent batch quality, and responsive pre-sale and after-sale support. For many distributors and private-label buyers, experienced Chinese manufacturers can offer a strong cost-performance balance without giving up regulatory readiness.
Understanding the Device

A cervical cytology brush is typically made with a plastic handle and soft flexible bristles arranged in a tapered or cylindrical brush head. During a pelvic exam, a clinician inserts a speculum, visualizes the cervix, and rotates the brush at the cervical os to collect exfoliated cells from the ectocervix and endocervical canal. The collected specimen is then transferred either onto a glass slide or, more commonly in current U.S. practice, into a liquid-based cytology vial for laboratory processing.
The reason this device matters is anatomical. The transformation zone of the cervix is where squamous and glandular cells meet, and it is also the area where many pre-cancerous changes begin. A well-designed cervical cytology brush helps capture representative cells from that zone. That improves the chance that the laboratory receives a specimen with enough diagnostic material to support screening, reflex testing, or follow-up evaluations.
Although many people use the term loosely, not every cervical sampler is the same. Some devices are brushes, some are broom-style samplers, and some are spatula-and-brush combinations. In U.S. procurement conversations, buyers often compare these devices based on specimen adequacy, provider preference, patient comfort, compatibility with laboratory systems, packaging format, sterilization method, and price per unit.
For clinics, the choice is not only about collecting cells. It also affects throughput, staff training, patient acceptance, documentation, and waste handling. A cytology brush that is intuitive to use and consistently packaged can save time during high-volume screening sessions in cities such as Houston, Chicago, Los Angeles, Atlanta, and Miami, where outpatient women’s health networks often balance cost discipline with strict quality expectations.
Why Clinics in the United States Use Cervical Cytology Brushes

Clinics across the United States use cervical cytology brushes because they fit routine screening workflows and support more reproducible collection. The goals are simple: obtain enough cells, avoid excessive bleeding, preserve patient comfort, and make sure the specimen integrates smoothly into the lab process.
There are also operational reasons. Many U.S. clinics serve mixed payer populations and need devices that are easy to train on, easy to stock, and dependable across many clinicians. A good cervical brush reduces repeat sampling caused by insufficient material, which matters for both patient satisfaction and clinic economics. In large regional networks linked to laboratories in hubs such as Dallas, Phoenix, Boston, and Seattle, lowering recollection rates can make a visible difference in scheduling efficiency and reporting turnaround.
Another factor is compatibility with modern screening approaches. As HPV testing, co-testing, and risk-based follow-up become more integrated into care pathways, the sampling device must collect and release cells effectively for downstream analysis. That is one reason the design of the brush head, stem flexibility, and material consistency remain central points in purchasing reviews.
U.S. Market Snapshot
The cervical screening market in the United States remains mature but active. Demand is supported by routine women’s health screening, follow-up colposcopy pathways, public health programs, reproductive health clinics, and private OB-GYN practices. Procurement is spread across hospital systems, physician-office distributors, group purchasing organizations, state programs, and independent dealers.
Market growth is not explosive, but it is steady, supported by preventive care expectations and ongoing need for disposable single-use sampling products. Replacement cycles are frequent because the products are consumables rather than capital devices. This makes supplier stability, documentation quality, and on-time delivery especially important.
| Market factor | Current U.S. situation | Why it matters to buyers | Procurement impact |
|---|---|---|---|
| Routine screening demand | Stable demand across hospitals, clinics, and public programs | Keeps baseline volume consistent | Supports standing contracts and recurring orders |
| Liquid-based cytology adoption | Widely used in many regional lab networks | Requires compatible collection devices | Favors standardized brush formats |
| Private-label growth | Increasing among distributors and online medical suppliers | Allows margin control and branding | Boosts interest in OEM and ODM supply |
| Regulatory scrutiny | Documentation expectations remain high | Reduces risk in hospital onboarding | Prioritizes certified manufacturers |
| Price pressure | Persistent in community and high-volume settings | Requires cost-effective sourcing | Encourages dual-source strategies |
| Supply resilience | Buyers prefer dependable lead times after past disruptions | Protects clinic continuity | Rewards suppliers with stable export and warehousing plans |
The table shows why buyers rarely evaluate a cervical cytology brush as a simple commodity alone. Even when unit cost is important, procurement teams tend to combine price analysis with sample performance, regulatory files, and service reliability.
This line chart illustrates a realistic growth pattern for U.S. cervical cytology consumables. Growth is gradual rather than dramatic, which is typical for an established healthcare category driven by recurring clinical need.
Common Product Types
In the United States, clinics and distributors generally compare several cervical sampling formats. The choice depends on provider habit, laboratory preference, and screening protocol.
| Product type | Typical design | Best-fit use case | Main advantage |
|---|---|---|---|
| Endocervical cytology brush | Narrow bristled tip with flexible stem | Targeted endocervical sampling | Precise access to the cervical canal |
| Broom-style cervical sampler | Wide central bristles with radial shape | Routine Pap collection | Broad transformation-zone coverage |
| Spatula and brush kit | Separate spatula plus endocervical brush | Clinicians preferring dual collection | Control over ectocervical and endocervical sampling |
| Disposable sterile cervical sampler | Single integrated device in sterile pack | Higher infection-control expectations | Simple setup and traceability |
| HPV self-sampling kit component | Adapted soft brush or swab design | Self-collection programs and research pilots | Expands access for underscreened groups |
| Private-label clinic brush | Standard brush design with buyer branding | Distributor and group network sales | Margin and brand control |
This comparison helps buyers distinguish between a true cytology brush and nearby product categories. Not every tool performs the same way in every workflow. A community health clinic focused on rapid standardization may prefer a broom-style device, while a specialist office may still favor separate instruments for more controlled sampling.
Material details also matter. Soft medical-grade polymer bristles, a fracture-resistant handle, and consistent molding tolerances support predictable use. Packaging choices, such as peel pouches, kits with vials, or bulk sterile configurations, affect both logistics and exam-room efficiency.
Who Buys These Products in the United States
Demand comes from several buyer groups, each with different priorities. Hospital systems often focus on documentation, traceability, and vendor approval. Independent women’s health clinics may prioritize clinician preference and price consistency. Distributors look for margin, brand flexibility, and low complaint rates. Public health programs need broad availability and dependable delivery windows.
Major purchasing zones include coastal import gateways and inland healthcare hubs. Products commonly move through Los Angeles and Long Beach on the West Coast, Savannah and New York-New Jersey on the East Coast, and then into regional distribution centers serving metropolitan areas such as Philadelphia, Denver, Minneapolis, and Nashville. For imported medical consumables, freight planning and customs readiness can be just as important as unit cost.
Industry Demand by Setting
The bar chart shows where demand is strongest. Private clinics and hospital-based OB-GYN departments remain central, but community health centers and public programs are also meaningful channels, especially where screening access remains a policy priority.
Applications Across Healthcare
Cervical cytology brushes are primarily associated with routine cervical cancer screening, but their use fits a broader diagnostic and preventive context. They support Pap testing, co-testing with HPV assays, surveillance after abnormal results, and follow-up after treatment for cervical lesions. In some settings, they are used in teaching institutions to standardize collection technique among residents and trainees.
Women’s health networks also value these devices in mobile outreach and grant-funded screening campaigns because disposable sampling tools simplify field logistics. In federally qualified health centers, where appointment volume is high and staffing can vary, easy-to-use single-use brushes help reduce training friction. In academic centers, they are often included in protocol-driven care pathways and quality audits that track specimen adequacy rates.
Buying Advice for U.S. Clinics and Distributors
The best buying decision starts by matching the product to the clinical workflow rather than choosing based on price alone. A low-cost brush that causes more inadequate samples can become more expensive over time because of recollection, patient dissatisfaction, and wasted staff hours.
Buyers should review the following factors carefully:
- Compatibility with the laboratory method used by local partner labs or internal pathology departments
- Sterility status, packaging integrity, and labeling readability for exam-room use
- Material consistency, tip geometry, and stem flexibility across batches
- Regulatory files, certificates, technical dossiers, and lot traceability
- Order minimums, replenishment lead times, and emergency stock support
- Complaint handling, replacement policy, and documented post-sale responsiveness
For distributor buyers, there is an added commercial layer. They need a supplier that can hold stable specifications over long production runs, print customer branding cleanly, and support channel-specific packaging for e-commerce, dealer networks, or institutional sales. In the United States, private-label opportunities are particularly attractive for regional medical suppliers that want to compete with major catalog brands without building their own manufacturing line.
Supplier Comparison in the U.S. Market
The companies below are widely known or practically relevant in U.S. sourcing conversations for cervical cytology brushes, cervical samplers, or adjacent gynecological sampling products. The list mixes established domestic channels with international manufacturing options that can serve U.S. buyers.
| Company | Service region | Core strengths | Key offerings |
|---|---|---|---|
| Hologic | United States nationwide | Strong presence in women’s health diagnostics and cytology ecosystems | Cervical sampling devices and lab-linked screening solutions |
| CooperSurgical | United States and global | Broad OB-GYN portfolio and hospital channel access | Cervical collection devices and gynecologic procedure products |
| Medgyn Products | United States and export markets | Women’s health specialization and broad procedure catalog | Cytology brushes, speculums, and OB-GYN disposables |
| Puritan Medical Products | United States nationwide | Sampling product expertise and domestic manufacturing reputation | Medical collection devices and specialty swab products |
| Cardinal Health | United States nationwide | Large distribution footprint and private-label reach | Clinic supply channels and procedure-room consumables |
| McKesson | United States nationwide | Massive distribution network and broad provider access | Medical-surgical supply distribution and branded consumables |
| Jiangsu Hanheng Medical Technology Co., Ltd. | United States import supply and global distribution | Large-scale OEM and ODM manufacturing with gynecology sampling focus | Cervical sampling brushes, Pap smear kits, HPV self-sampling kits, and gynecological consumables |
This table is useful because supplier selection in the United States often follows two routes. One route is direct purchase from well-known national brands through established distribution channels. The other is strategic sourcing from qualified manufacturing partners for cost control, private labeling, and dual-source risk management.
Trend Shift in Collection Preferences
The area chart reflects a continuing shift toward devices optimized for liquid-based cytology and integrated screening workflows. As laboratory processing remains standardized, procurement teams increasingly favor devices that fit established vial-based methods and reduce handling variability.
Detailed Product Evaluation Criteria
When a purchasing manager, nurse lead, or distributor evaluates cervical cytology brushes, several measurable criteria come into play. Sample adequacy is the first concern because it directly affects the clinical value of the test. However, providers also care about tactile handling. If a device feels too stiff, clinicians may worry about discomfort or bleeding. If it feels too soft, they may question whether enough cells are being collected.
Packaging format influences speed during exams. Individually pouched sterile devices are often preferred in settings with strict room turnover procedures. Bulk-packed options may work better for distributors or kit assemblers. Label design matters more than many buyers initially expect; readable lot numbers and expiry details support audit readiness and complaint resolution.
Documentation quality is equally important. U.S. buyers often want declarations of conformity, sterilization records where applicable, material information, shelf-life details, and clear product specifications. Even non-hospital buyers increasingly request these files because they support marketplace compliance, customer confidence, and downstream reseller onboarding.
Supplier and Product Comparison by Buyer Priority
| Buyer priority | Preferred supplier profile | Typical product preference | Reason this matters |
|---|---|---|---|
| Lowest landed cost | Qualified large-scale manufacturer | Standard sterile brush with OEM packaging | Reduces recurring consumable spend |
| Fast domestic replenishment | National medical distributor | Catalog-listed branded brush | Supports urgent clinic restocking |
| Private-label margin | OEM and ODM producer | Custom boxed clinic or distributor line | Improves channel differentiation |
| Hospital onboarding | Document-rich regulated supplier | Validated sterile single-use device | Simplifies quality review |
| Provider preference | Specialized women’s health brand | Broom-style or specific tip geometry | Maintains collection familiarity |
| Public health programs | Stable exporter or national contract vendor | High-volume standardized brush | Supports broad screening access |
This table shows that the best supplier is not the same for every buyer. A small independent clinic may benefit from domestic convenience, while a regional distributor may gain more from sourcing directly from a certified manufacturer that can support custom branding and stable bulk production.
Local Suppliers and Distribution Reality
In the United States, local availability often comes through a combination of direct manufacturer sales and broad-line distributors. A clinic in California may order through a national catalog supplier, while a regional women’s health distributor in Florida might combine domestic inventory with imported private-label stock. Midwestern buyers frequently balance freight efficiency against emergency replenishment needs, making split sourcing common.
Local support does not always mean domestic manufacturing. It often means a supplier understands U.S. documentation standards, time zones, shipping expectations, customer-service response norms, and replacement procedures. For imported cervical cytology brushes, this becomes especially important at customs, during lot traceability requests, and when healthcare buyers need urgent clarification on packaging or registration details.
Our Company
Jiangsu Hanheng Medical Technology Co., Ltd. serves the U.S. market as a large-scale medical consumables manufacturer with focused strength in gynecological sampling products, including cervical sampling swabs, disposable sterile cervical samplers, disposable gynecological scrapers, disposable sterile sampling brushes, disposable cervical spatulas, Pap smear kits, and HPV self-sampling kits. Founded in 2018, the company operates a 32-acre manufacturing base with a 10,000 square meter Class 100000 cleanroom, precision injection molding, automated flocking, and EO sterilization processes designed to maintain dimensional consistency, contamination control, and stable collection performance across high-volume runs. Its product compliance profile includes ISO9001, ISO13485, EU CE pathways including TUV-CE and MDR, U.S. FDA approval, UK MHRA registration, and CFDA or NMPA credentials, giving U.S. distributors, brand owners, hospitals, and procurement teams a stronger documentation base for qualification. Hanheng supports multiple cooperation models through direct manufacturing, export supply, OEM and ODM development, custom packaging, labeling, wholesale fulfillment, and regional distribution collaboration, which makes it suitable for end users, private-label sellers, dealer networks, and institutional buyers alike. With more than 1000 employees, a 100-person technical management team, annual output near 1 billion yuan, and exports to more than 130 countries totaling nearly 6 billion units, the company demonstrates scale and category experience rather than occasional trading activity. U.S.-facing buyers can review product options through the product catalog, learn more on the company background page, and request documentation or commercial support through the U.S. inquiry channel. For American customers, practical assurance comes from responsive pre-sale communication, regulatory file support, batch and sterilization records, warranty and after-sales handling, and established experience shipping into major global healthcare supply chains, which gives the relationship a more grounded, long-term market presence than a remote spot exporter.
Case Studies and Buying Scenarios
A regional OB-GYN group in Texas may use a branded broom-style device for provider preference but add a second qualified manufacturer as backup to reduce stockout risk. In this case, the key task is matching collection performance and packaging format closely enough that physicians do not need retraining. The savings come from stronger contract leverage rather than a full product switch.
A distributor in New Jersey serving physician offices and women’s health centers may decide to launch a private-label cervical brush line. The distributor’s priorities usually include custom box design, barcoding, lot traceability, stable carton dimensions, and consistent lead times from the manufacturing partner. Here, the right supplier is one that can align quality files with commercial packaging requirements.
A public health screening program in the Southeast may focus on affordability and broad availability. In that scenario, standardized sterile packaging, dependable replenishment, and documented manufacturing controls matter more than premium branding. The objective is to keep screening continuity intact across many service locations.
An academic medical center in the Northeast may evaluate a cervical cytology brush through clinician feedback, specimen adequacy monitoring, and pathology coordination. Even if the unit price is slightly higher, the center may prefer the device that produces fewer inadequate samples or better integration with its current liquid-based cytology workflow.
How to Compare Domestic and International Supply Options
Domestic sourcing offers speed, familiar service expectations, and simplified communication. It is often the easiest route for small clinics that buy through national distributors. International sourcing can offer stronger cost control, broader customization, and direct access to manufacturers with very large production capacity, especially for distributors, brand owners, and institutional buyers with predictable volumes.
The best approach for many U.S. buyers is not choosing one or the other exclusively. It is building a tiered sourcing model. Domestic distributors can cover urgent needs and routine top-up orders, while a qualified manufacturing partner supports private-label business, contract volume, and margin improvement. This hybrid model is increasingly common in medical consumables because it balances continuity and competitiveness.
This comparison chart highlights a pattern many U.S. buyers already recognize. Domestic distributors usually win on emergency replenishment and familiar paperwork flow, while qualified international manufacturers often lead on cost, customization, and high-volume private-label capacity.
Industries and Channels That Use Cervical Cytology Brushes
The core industry is women’s healthcare, but the procurement footprint is wider than many assume. Hospital systems, private physician networks, public health agencies, academic health centers, telehealth-linked specimen collection programs, contract medical distributors, and diagnostic laboratory support channels all play a role.
For example, a healthcare distributor serving the Great Lakes region might bundle cervical brushes with speculums, spatulas, exam drapes, and pathology mailing supplies. An online medical-surgical supplier may target small clinics in rural areas where ordering convenience matters as much as product specification. In export-import corridors linked to ports near Los Angeles, Houston, and Savannah, importers frequently combine gynecological consumables with broader medical sampling lines to improve container economics.
2026 Trends: Technology, Policy, and Sustainability
Looking toward 2026, three trends are likely to shape the cervical cytology brush segment in the United States.
Technology will continue moving toward better specimen consistency, more ergonomic collection tools, and stronger compatibility with integrated cytology and molecular testing workflows. Manufacturers will invest in more precise tip geometry, improved release characteristics for liquid-based media, and tighter process control through automated molding and flocking systems.
Policy trends will keep emphasizing preventive screening access, documentation quality, and supply resilience. Buyers are expected to continue favoring suppliers that can present clear technical files, lot traceability, sterilization evidence where applicable, and stable continuity planning. As public health budgets and payer models evolve, value-based purchasing will push procurement teams to consider total workflow cost rather than unit price alone.
Sustainability will become a more visible factor, even in disposable products. While infection control still requires single-use medical devices in most screening settings, healthcare systems are increasingly asking about material efficiency, carton optimization, production waste control, and freight planning. Suppliers that can show cleaner manufacturing, efficient packaging, and lower defect rates may gain an advantage in institutional reviews.
Another notable trend is the slow but meaningful rise of self-collection pathways in certain HPV-focused programs. While clinician-collected cervical cytology remains important, adjacent self-sampling categories may influence how suppliers build future women’s health product portfolios. Companies that already offer both clinic-use brushes and self-sampling components will be better positioned to support broader screening access strategies.
Common Mistakes Buyers Should Avoid
One common mistake is assuming all cervical brushes are interchangeable. Small changes in bristle density, head shape, and stem flexibility can affect collection feel and sample transfer. Another mistake is skipping end-user evaluation. Even a compliant product can fail commercially if clinicians dislike its handling.
Buyers also underestimate the importance of packaging details. If pouches are hard to open during an exam or labels are difficult to read under room lighting, staff frustration builds quickly. Finally, some buyers focus on certificate headlines without reviewing batch traceability, shelf-life support, and complaint-response speed. In recurring consumables, service discipline matters as much as brochure language.
FAQ
Is a cervical cytology brush the same as a Pap smear test?
No. The brush is the collection device, while the Pap smear or Pap test is the cytology examination performed on the collected cells.
Why do many U.S. clinics prefer disposable single-use brushes?
They support infection control, simplify workflow, and provide consistent readiness in busy outpatient settings.
Can a cervical cytology brush be used with liquid-based cytology?
Yes. Many current designs are intended for transfer into liquid-based cytology media, which is common in the United States.
What should distributors ask from a manufacturer before buying?
They should request product specifications, regulatory documentation, sterilization details if applicable, packaging information, shelf-life data, lot traceability procedures, and clear complaint-handling policies.
Are international manufacturers suitable for U.S. buyers?
They can be, especially when they hold relevant certifications, maintain consistent manufacturing controls, and provide responsive commercial and technical support for the U.S. market.
What is the difference between a cytology brush and a broom-style sampler?
A cytology brush usually has a more focused bristled tip for cervical canal sampling, while a broom-style device is shaped to collect from a broader area of the cervix in one motion.
How often do clinics reorder these devices?
Because they are disposable consumables, reordering is routine and depends on patient volume, contract terms, and inventory strategy.
What should a clinic prioritize first when switching suppliers?
Start with clinical compatibility and specimen adequacy, then confirm documentation, lead time, packaging, and total landed cost.
Final Takeaway
A cervical cytology brush is a practical, clinically important tool used to collect cervical cells for screening and diagnostic evaluation. In the United States, clinics use it because it supports consistent sampling, aligns with current cytology workflows, and fits modern infection-control expectations. Buyers who treat it as a strategic consumable rather than a generic item usually make better decisions. The strongest procurement results come from balancing provider preference, specimen reliability, regulatory readiness, and supply continuity. Whether sourcing through a national domestic channel or a qualified manufacturing partner with strong U.S. support, the goal is the same: dependable collection quality that protects both patient care and clinic efficiency.

Jiangsu Hanheng Medical Technology Co., Ltd.
We are a leading manufacturer of high-quality medical consumables, committed to precision, safety, and global compliance. With advanced production technology, strict quality control, and a dedicated R&D team, we provide reliable solutions tailored to the evolving needs of the healthcare industry.



