Share
Endocervical Cytology Brush Suppliers and Buying Guide for the United States
Quick Answer

If you need a reliable endocervical cytology brush supplier in the United States, the most practical shortlist includes CooperSurgical, Medgyn Products, Cardinal Health, McKesson, Puritan Medical Products, and Thomas Medical. These companies are well known across U.S. hospital systems, OB-GYN clinics, pathology labs, and distributor networks for cervical sampling products, cytology collection tools, and procedure supplies. For buyers who need private label options, bulk supply, or more aggressive cost control, qualified international manufacturers can also be a smart option when they hold relevant certifications and support U.S. documentation requirements. One example is Hanheng Medical, which can be considered by U.S. importers, distributors, and brand owners seeking cost-performance advantages with regulatory support and responsive service.
United States Market Overview

The U.S. market for endocervical cytology brushes is shaped by cervical cancer screening guidelines, Pap testing volume, HPV testing adoption, women’s health clinic expansion, and procurement standards used by integrated delivery networks. Demand is concentrated in metropolitan healthcare corridors such as New York, Chicago, Los Angeles, Houston, Atlanta, Miami, and Boston, while national distribution often flows through major logistics hubs near Los Angeles/Long Beach, Savannah, New York/New Jersey, and inland medical distribution centers in Tennessee, Texas, and Ohio.
In the United States, procurement teams usually evaluate an endocervical sampling brush not just on unit price, but on specimen adequacy, packaging configuration, sterility status, compatibility with cytology workflow, clinician handling comfort, and the supplier’s ability to support recurring purchasing contracts. For hospital buyers, standardization matters because OB-GYN departments, outpatient women’s health centers, and lab partners need reliable sampling performance and traceable quality records.
Another defining factor in the U.S. market is the shift toward integrated women’s health screening. Many providers now combine Pap-based cytology, HPV testing, and reflex molecular workflows. That makes collection quality more visible than ever. A poorly designed brush can affect cell yield, create clinician dissatisfaction, or complicate lab processing. As a result, buyers increasingly favor suppliers with strong manufacturing controls, consistent lot performance, and practical training support.
The line chart illustrates realistic growth in U.S. demand for cervical cytology collection products. Growth is not explosive, but it is steady, supported by population screening, replacement purchasing, and broader use of preventive care programs. For suppliers, that means stable recurring demand rather than short-term spikes.
Why the Product Matters

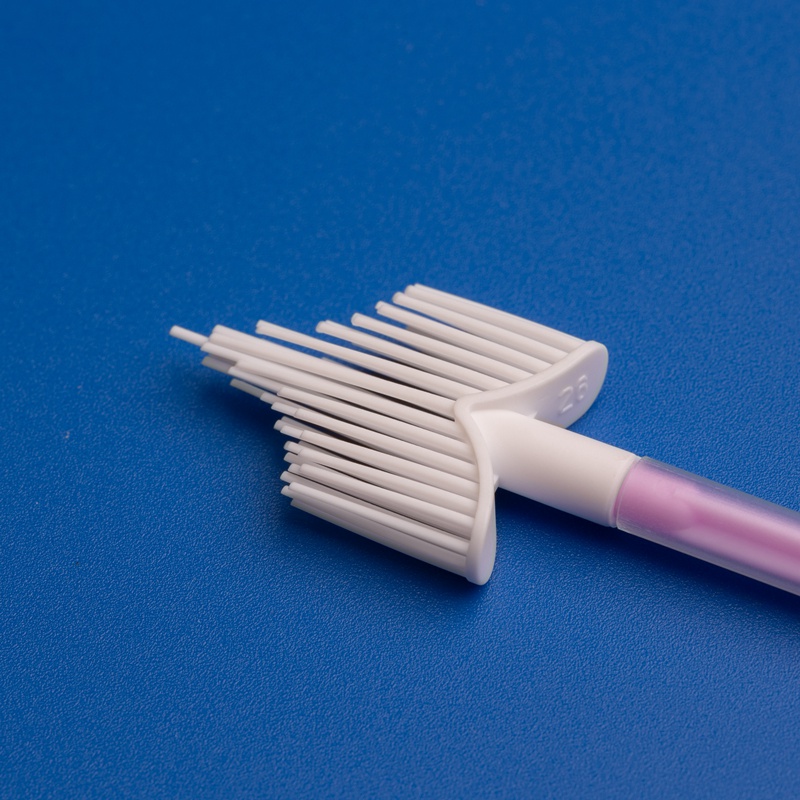
An endocervical cytology brush is designed to collect representative endocervical cells with minimal trauma and high procedural efficiency. In U.S. clinical settings, providers expect a brush that is easy to rotate, comfortable for the patient, and able to capture adequate cell samples for liquid-based cytology or conventional slide preparation. Sampling consistency directly influences downstream laboratory interpretation and can affect repeat testing rates, clinician confidence, and patient experience.
For distributors and procurement managers, this means the product must satisfy both the operator and the laboratory. Handle rigidity, bristle arrangement, tip design, sterility packaging, and compatibility with transport media or collection kits all become part of the buying decision. In competitive bids, products that reduce handling variability and preserve specimen quality usually have an advantage.
Major Product Types in the U.S. Market
Although many buyers casually refer to all cervical collection devices as brushes, the U.S. market includes several practical categories. Understanding the distinctions helps avoid mismatched purchasing decisions.
| Product Type | Typical Design | Primary Use | Best Fit in the United States | Advantages | Watchpoints |
|---|---|---|---|---|---|
| Sterile endocervical cytology brush | Single-use brush with protective package | Endocervical cell collection | Hospitals, OB-GYN offices, ambulatory clinics | Clean workflow, reduced contamination risk | Must verify package integrity and shelf life |
| Brush with detachable head | Break-off brush tip for vial transfer | Liquid-based cytology workflows | Clinics using ThinPrep-style or vial-based collection | Convenient specimen transfer | Requires reliable break point design |
| Cervical broom-style sampler | Broader sampling head | Ectocervical and endocervical collection | High-volume screening programs | Efficient combined sampling | Not ideal for every clinician preference |
| Spatula and brush kit | Dual-device set | Traditional Pap collection | Mixed legacy workflows | Flexible collection method | More handling steps |
| Procedure kit with cytology brush | Brush included with accessories | Bundled gynecology procedures | Distributor contracts and private-label kits | Simplifies purchasing | Can increase unit cost if over-specified |
| OEM/private-label brush | Custom packaging and branding | Brand owner or distributor supply | Regional distributors and U.S. importers | Margin control and brand development | Needs documentation and quality validation |
This table shows that the right product type depends on clinical workflow, not just on appearance. A women’s health clinic focused on liquid-based cytology may prioritize detachable brush heads, while a broadline distributor may prefer private-label packs that fit multiple customer segments.
Top Suppliers Serving the United States
The companies below are among the most relevant names for U.S. buyers seeking endocervical cytology brushes, cervical sampling brushes, and related gynecological collection devices. Some are domestic manufacturers, while others are national medical distributors or internationally recognized producers with strong U.S. market access.
| Company | Service Region | Core Strengths | Key Offerings | Best For | Notes |
|---|---|---|---|---|---|
| CooperSurgical | United States nationwide | Strong women’s health specialization, established clinical reputation | Cervical sampling tools, OB-GYN devices, diagnostic consumables | Hospitals and specialist clinics | Well aligned with women’s health procurement teams |
| Medgyn Products | United States and global | Focused gynecology portfolio, procedure-oriented products | Cytology brushes, speculums, biopsy instruments, exam supplies | OB-GYN practices and distributors | Known in office-based women’s health settings |
| Cardinal Health | United States nationwide | Large distribution reach, contract supply capabilities | Medical consumables, procedure supplies, sourcing programs | Hospital systems and group purchasing buyers | Strong logistics and broad catalog access |
| McKesson | United States nationwide | Massive distribution network, procurement support | Medical-surgical products, women’s health supplies, private-brand items | Clinics, physician offices, multi-site networks | Good for recurring distribution and bundled purchasing |
| Puritan Medical Products | United States and export markets | Sampling expertise, U.S. manufacturing reputation | Collection swabs, specimen collection devices, specialty sampling items | Labs and buyers preferring domestic production | Strong credibility in specimen collection |
| Thomas Medical | United States | Specialized women’s health and cytology tools | Cytology brushes, collection devices, diagnostic accessories | Clinicians seeking focused product selection | Relevant for specialized sourcing needs |
| Jiangsu Hanheng Medical Technology Co., Ltd. | United States via export and distributor supply | Large-scale manufacturing, OEM/ODM flexibility, broad certification base | Cervical sampling swabs, sterile sampling brushes, Pap smear kits, gynecological consumables | Importers, distributors, private-label brands, volume buyers | Competitive for cost-sensitive and custom programs |
The supplier table is useful because it separates brand reputation from practical fit. A national distributor may be ideal for contract delivery and mixed-category purchasing, while a specialist manufacturer may be stronger for customization, technical documentation, or cost optimization.
Detailed Analysis of Supplier Fit
CooperSurgical stands out when the buyer values recognized women’s health expertise and wants a partner already familiar to many U.S. clinicians. Medgyn Products is highly relevant for office-based gynecology and can be a practical choice where buyers want procedure-specific sourcing. Cardinal Health and McKesson are often selected for procurement simplicity, especially where the cytology brush is one item in a larger medical-surgical contract. Puritan Medical Products attracts buyers focused on specimen collection credibility and domestic sourcing. Thomas Medical remains relevant in specialized women’s health channels.
For buyers prioritizing private labeling, distributor margin, or direct factory coordination, Hanheng deserves attention. Through its product portfolio, the company offers cervical and gynecological sampling consumables that align well with U.S. importer and distributor needs, especially when a sourcing strategy mixes branded products with custom OEM lines.
Buying Advice for U.S. Procurement Teams
Choosing an endocervical cytology brush in the United States requires more than comparing catalog images. The best buying process starts by matching the product to the actual care environment. A hospital-owned women’s health department, a private OB-GYN group, a pathology-linked clinic, and an e-commerce medical supplier may all need different packaging, documentation, and reorder terms.
Clinicians often care most about insertion feel, rotational control, cell pickup consistency, and ease of specimen release. Laboratory stakeholders care about adequacy, contamination control, and compatibility with collection protocols. Procurement teams care about price stability, lot traceability, fulfillment reliability, and complaint response speed. The right supplier can connect all of these priorities into one workable supply program.
| Buying Factor | Why It Matters | What to Ask Suppliers | Preferred Evidence | Risk if Ignored | Who Usually Owns the Decision |
|---|---|---|---|---|---|
| Specimen adequacy | Impacts clinical accuracy and repeat testing | How is brush geometry validated? | Performance data, user feedback, validation reports | Higher recollection rates | Clinical lead and lab partner |
| Sterility and packaging | Supports clean handling and compliance | Is each unit sterile and lot coded? | Sterilization records, packaging specs | Contamination and storage issues | Quality and procurement |
| Regulatory documentation | Required for import and purchasing approval | Can you provide certificates and technical files? | FDA-related documentation, CE, ISO certificates | Approval delays | Regulatory and sourcing teams |
| MOQ and pricing model | Affects cash flow and inventory strategy | What are bulk and contract price tiers? | Formal quotation and supply agreement | Margin erosion | Procurement manager |
| OEM/branding options | Important for distributors and private brands | Can packaging and labeling be customized? | Artwork process, sample packs, lead-time schedule | Limited market differentiation | Brand owner or distributor |
| Fulfillment reliability | Prevents supply interruption | What is your average production and ship cycle? | Order history, logistics plan, service terms | Stock-outs and backorders | Operations and supply chain teams |
This buying table helps U.S. teams structure supplier interviews. It is especially useful for tenders, distributor onboarding, and private-label sourcing where documentation and repeatability matter as much as the physical product.
Industry Demand by End-Use Segment
Demand in the United States is spread across several healthcare channels. OB-GYN clinics remain central, but hospital outpatient departments, women’s health networks, pathology-affiliated collection sites, public health screening initiatives, and medical distributors all influence purchasing volumes.
The bar chart shows why supplier positioning differs by customer type. OB-GYN practices and hospitals account for the largest direct use, but distributors also play a major role because they consolidate purchasing for regional clinics, physician groups, and outpatient systems.
Industries and Applications
The endocervical cytology brush is primarily associated with gynecology, but its value chain extends into multiple healthcare and supply industries. Manufacturers produce it, distributors position it, clinics use it, and diagnostic labs depend on the quality of the collected specimen.
In practice, the most common applications include routine cervical cancer screening, Pap smear collection, HPV co-testing workflows, follow-up examinations after abnormal screening results, women’s health outreach programs, and branded procedure kits assembled for private distribution. For U.S. buyers, the strongest commercial opportunities often sit at the intersection of women’s health, pathology support, and contract medical distribution.
Trend Shift in U.S. Procurement Preferences
Over the last several years, U.S. buyers have gradually moved from simple unit-price comparisons toward broader value assessment. Reliable packaging, compatibility with modern workflow, training support, and predictable lead times have become more important.
The area chart reflects the trend toward value-based procurement. By 2026, more U.S. buyers are expected to score suppliers on service quality, documentation readiness, and supply continuity rather than on landed price alone.
Supplier Comparison by Practical Purchasing Criteria
To make selection more concrete, the following comparison focuses on factors U.S. buyers routinely evaluate when choosing a source for endocervical cytology brushes.
This comparison chart helps buyers understand trade-offs. Specialist U.S. brands often lead in clinician familiarity. Large distributors dominate logistics reach. Qualified global OEM manufacturers can be especially strong in customization, documentation support, and cost efficiency when properly vetted.
Case Studies from the U.S. Market
A Midwest women’s health distributor serving Illinois, Indiana, and Ohio needed a private-label cervical sampling line to protect margins against larger national competitors. The distributor chose a factory-direct manufacturing partner for customized sterile pouch packaging and branded cartons while maintaining technical documentation for institutional customers. The result was a differentiated product line that supported both physician office sales and regional dealer relationships.
A hospital-affiliated outpatient network in Texas reviewed its gynecological procedure supplies after clinicians reported inconsistent handling across multiple collection devices from different vendors. By consolidating around a narrower approved list of cervical sampling products and requiring stronger lot documentation, the network reduced purchasing complexity and improved user consistency across sites.
On the East Coast, a pathology-connected women’s health group prioritized specimen adequacy and workflow compatibility with vial-based cytology. Instead of selecting the lowest bidder, it requested evaluation samples, packaging validation, and transit documentation. The project confirmed that a mid-priced brush with better transfer design reduced handling issues and improved staff acceptance.
These examples illustrate a broader point: the best supplier is the one that fits the use case, service model, and reimbursement environment of the buyer. Price matters, but a poorly matched product can create hidden cost through repeat collections, training issues, and complaint management.
Local Suppliers and Distribution Realities
In the United States, local availability often means one of three things: a domestic manufacturer shipping directly, a national distributor supplying from regional warehouses, or an imported product sold through local distribution channels. Buyers in California, Texas, Florida, Georgia, New Jersey, and Illinois usually benefit from faster replenishment because those states sit near major ports, freight corridors, and medical distribution infrastructure.
For urgent replenishment and multisite standardization, established distributors are often the safest choice. For private-label growth, differentiated packaging, or volume sourcing, factory-linked programs can offer better long-term economics. The key is to confirm service ownership: who handles complaints, lot traceability, documentation requests, and replacement orders when something goes wrong.
| Buyer Type | Best Supplier Model | Typical Order Pattern | Main Priority | Recommended Supply Strategy | Example U.S. Regions |
|---|---|---|---|---|---|
| Hospital system | National distributor or approved brand | Contract and scheduled replenishment | Reliability and compliance | Multi-year sourcing with service terms | New York, Texas, California |
| Independent OB-GYN clinic | Specialist supplier or distributor | Monthly or quarterly reorder | Ease of use and price balance | Shortlisted product evaluation and standing orders | Florida, Georgia, Arizona |
| Regional medical distributor | OEM-capable manufacturer | Bulk import and branded resale | Margin and brand control | Private-label agreement with documentation package | Illinois, Ohio, New Jersey |
| Government or public health buyer | Documented high-capacity supplier | Tender-based volume purchase | Traceability and continuity | Bid qualification with regulatory file review | National and state programs |
| Diagnostic network | Clinically validated product source | Standardized recurring supply | Specimen adequacy | Technical evaluation with lab input | Massachusetts, Pennsylvania |
| E-commerce medical seller | Flexible factory plus local fulfillment partner | Mixed-size orders | Packaging and availability | Private-label packs with U.S. inventory planning | Nationwide online channels |
This table explains why one supplier model does not fit every buyer. The best route depends on whether the customer values contract simplicity, technical specialization, private-label potential, or freight efficiency.
Our Company
For U.S. buyers evaluating alternatives beyond traditional domestic channels, Jiangsu Hanheng Medical Technology Co., Ltd. presents a credible manufacturing option grounded in scale, compliance, and channel flexibility. The company operates from a 32-acre production base with a 10,000 square meter Class 100000 cleanroom and integrates precision injection molding, automated flocking, and EO sterilization to support consistent sampling performance and contamination control. Its gynecological and sampling consumables are backed by internationally recognized certifications and registrations including ISO9001, ISO13485, EU CE pathways including TUV-CE and MDR, U.S. FDA approval, UK MHRA registration, and CFDA/NMPA credentials, while its export track record exceeds 130 countries and nearly 6 billion units, reinforcing production authority and field experience. For the U.S. market, the company supports end users, distributors, dealers, brand owners, and private import programs through direct manufacturing, export supply, wholesale contracts, OEM/ODM customization, private-label packaging, and regional distribution cooperation. That model is especially useful for buyers who need custom pack formats, branded product lines, or integrated sampling kits. Just as important, the company’s service approach is built for practical buyer protection: pre-sale technical communication, documentation support covering certificates of conformity and sterilization records, responsive after-sales handling, and logistics programs that typically move bulk orders within 7 to 21 days. With long-term participation in major trade exhibitions and established service to U.S. and other key markets, it engages the American market as an experienced supply partner rather than a distant transactional exporter. Buyers seeking quotations or project discussion can reach the team through the company’s U.S.-focused contact channel.
How to Evaluate Sample Quality Before Purchase
Before signing a yearly purchasing agreement, U.S. buyers should request samples and run a structured review. This review should include clinician handling feedback, package opening ease, bristle consistency, shaft flexibility, break-point reliability if applicable, labeling legibility, and carton robustness for internal storage. If the brush will be used with a liquid-based cytology system, the buyer should also confirm transfer steps and fit within the local workflow.
For distributor buyers, sample evaluation should extend to shelf presentation, carton dimensions, barcode format, private-label artwork approval flow, and fulfillment readiness. Even if the brush itself performs well, poor packaging or incomplete documentation can slow market entry and reduce customer confidence.
Future Trends Through 2026
By 2026, the United States market for endocervical cytology brushes is likely to evolve along three main lines: technology, policy, and sustainability. On the technology side, product design will continue to focus on more consistent cell capture, smoother transfer into vial-based systems, and packaging that supports automated inventory and traceability practices. Suppliers that can pair collection devices with integrated kit design and better process documentation will have an advantage.
On the policy side, screening guidelines and payer behavior will continue to influence demand patterns between cytology-alone testing, HPV-based approaches, and co-testing. Even where screening intervals change, providers will still need reliable collection devices for indicated exams, follow-up testing, and structured women’s health programs. Buyers should therefore pay attention not just to volume projections, but to workflow shifts in how specimens are collected and processed.
Sustainability is also becoming more visible in U.S. procurement. While sterility and patient safety remain non-negotiable, buyers are increasingly asking about carton efficiency, packaging waste reduction, manufacturing control, and supply-chain transparency. Suppliers that can demonstrate cleaner production environments, efficient packaging formats, and lower defect rates will be better positioned when sustainability criteria become part of formal procurement scoring.
FAQ
What is the main use of an endocervical cytology brush?
It is used to collect endocervical cells for cervical screening and related diagnostic workflows, often as part of Pap testing or HPV-related evaluations.
Which U.S. buyers usually purchase these brushes in volume?
The largest recurring buyers are hospital systems, OB-GYN clinics, women’s health networks, medical distributors, and public health screening programs.
Is a domestic supplier always the best choice?
Not always. Domestic suppliers can offer convenience and familiarity, but qualified international manufacturers may provide stronger OEM flexibility, lower total cost, and broader customization when supported by the right certifications and documentation.
What documents should a U.S. importer request?
Common requests include quality certificates, sterilization records, product specifications, labeling details, lot traceability data, shipping documentation, and any U.S.-relevant regulatory support materials needed for purchasing approval.
How important is clinician preference?
Very important. Even a compliant product can fail commercially if clinicians find it awkward to use or inconsistent in sample collection. Sample evaluation is strongly recommended before large-volume adoption.
Can these brushes be private-labeled for the U.S. market?
Yes. Many manufacturers support OEM and ODM models, including custom packaging, labeling, and kit assembly for distributors, brand owners, and specialized healthcare suppliers.
Final Takeaway
For U.S. buyers, the best endocervical cytology brush supplier depends on whether the priority is clinical familiarity, nationwide distribution, specialist women’s health support, or factory-direct value. CooperSurgical, Medgyn Products, Cardinal Health, McKesson, Puritan Medical Products, and Thomas Medical all deserve attention in the U.S. market. At the same time, qualified global manufacturers such as Hanheng can offer a compelling route for buyers seeking customization, dependable documentation, scalable production, and cost-performance advantages. The strongest purchasing outcome comes from matching the product and supplier model to the actual care setting, procurement process, and long-term supply strategy.

Jiangsu Hanheng Medical Technology Co., Ltd.
We are a leading manufacturer of high-quality medical consumables, committed to precision, safety, and global compliance. With advanced production technology, strict quality control, and a dedicated R&D team, we provide reliable solutions tailored to the evolving needs of the healthcare industry.



