Top Vaginal Brush Wholesalers for the European Market 2025
Share
1. Introduction: The Growing Demand for Vaginal Brushes in Europe
Over the past decade, the European medical consumables market has experienced rapid growth—especially in the field of women’s health. Among the most in-demand products are vaginal brushes, which are essential tools for collecting cervical or vaginal samples for diagnostic testing, including HPV screening and cervical cancer detection. As early detection becomes a healthcare priority across EU nations, hospitals, diagnostic labs, and women’s health clinics are seeking reliable suppliers for sterile, high-performance vaginal brushes at scale.
In 2025, the demand for vaginal brushes in Europe is projected to increase significantly due to:
- National cervical cancer screening programs (e.g., NHS Cervical Screening Programme in the UK, ZORA programme in Germany)
- The rise in self-sampling initiatives for HPV testing
- Expansion of women’s health clinics and diagnostic centers
- Growing awareness of preventive gynecological care
This surge in demand creates a lucrative opportunity for wholesalers, distributors, and procurement managers to partner with trusted suppliers who can meet volume requirements while maintaining strict EU regulations for medical devices.
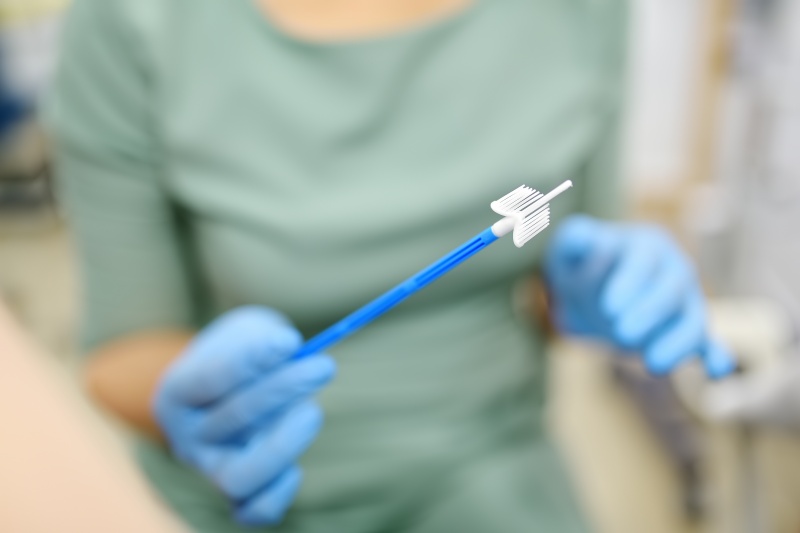
What Are Vaginal Brushes Used For?
Vaginal brushes, also known as cervical sampling brushes or gynecological sampling tools, are primarily used to:
- Collect epithelial cells from the cervix or vaginal wall
- Aid in the early detection of cervical cancer
- Assist in HPV DNA and STI testing
- Support cytology and microbiological analysis
These devices are typically used in:
- Gynecological examinations
- Lab and diagnostic settings
- National screening programs
- At-home self-sampling kits
Medical-grade vaginal brushes must meet rigorous standards for sterility, patient comfort, and precision sampling. As such, choosing the right wholesale supplier is critical for any organization involved in women’s health diagnostics.
2. Market Trends: Women’s Health & Diagnostic Consumables in 2025
The vaginal brush market is part of the broader women’s diagnostic consumables sector, which is undergoing transformation across the EU. Here’s a breakdown of the key trends driving the market in 2025:
Key EU Market Drivers
| Trend | Description |
|---|---|
| Cervical Cancer Screening Policies | Countries like France, Germany, and the UK are expanding public health initiatives to increase screening coverage. |
| Self-Sampling Kits | Women are increasingly opting for at-home HPV testing kits, which include sterile vaginal brushes. |
| EU MDR Regulations | The European Medical Device Regulation (MDR) enforces stricter quality and labeling standards, influencing supplier selection. |
| Preference for Disposable Devices | Infection control protocols post-COVID have driven demand for single-use, sterile consumables. |
| Digitalization of Healthcare | Increased use of telehealth and remote diagnostics supports the growth of mail-in sample collection tools. |
Market Size & Forecast
According to industry estimates:
- The European women’s diagnostic consumables market is projected to reach €1.2 billion by 2025.
- Vaginal brushes make up a growing sub-segment, with an expected CAGR of 7.8%.
- Germany, France, Italy, and the UK are the largest buying markets in the region.
Buyer Segments
| Buyer Type | Common Needs |
|---|---|
| Public Health Agencies | Bulk procurement via tenders, CE/FDA compliance |
| Hospitals & Clinics | Regular restocking, compatibility with lab systems |
| Self-Sampling Kit Brands | Custom packaging, private labeling, sterile brushes |
| Medical Distributors | Reliable supply chains, competitive wholesale pricing |
Understanding these buyer personas is critical for suppliers aiming to enter or expand in the European market.

.jpg)
3. Key Criteria for Choosing a Reliable Vaginal Brush Supplier
For B2B buyers, selecting a vaginal brush manufacturer or wholesaler isn’t just about price—it’s about consistent quality, regulatory compliance, and reliable logistics. Below are the key factors to consider:
A. Certification & Regulatory Compliance
Ensure the supplier meets international and EU standards:
- CE Marking (Essential for EU market entry)
- ISO 13485: Medical Device Quality Management System
- FDA Registration (if importing from the US or China)
- Sterilization Validation Reports (e.g., Gamma Ray Sterilization)
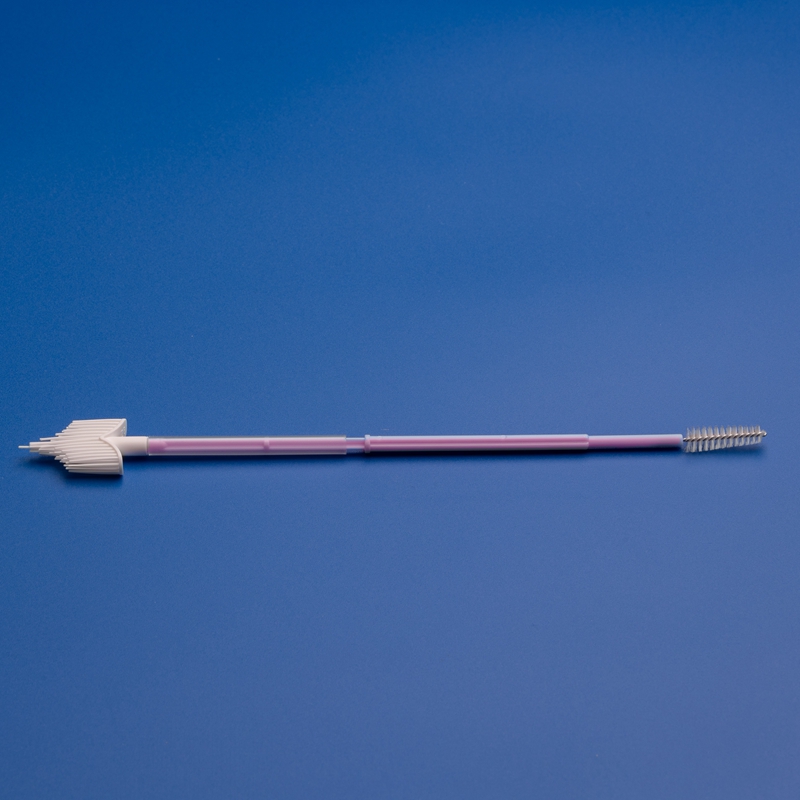
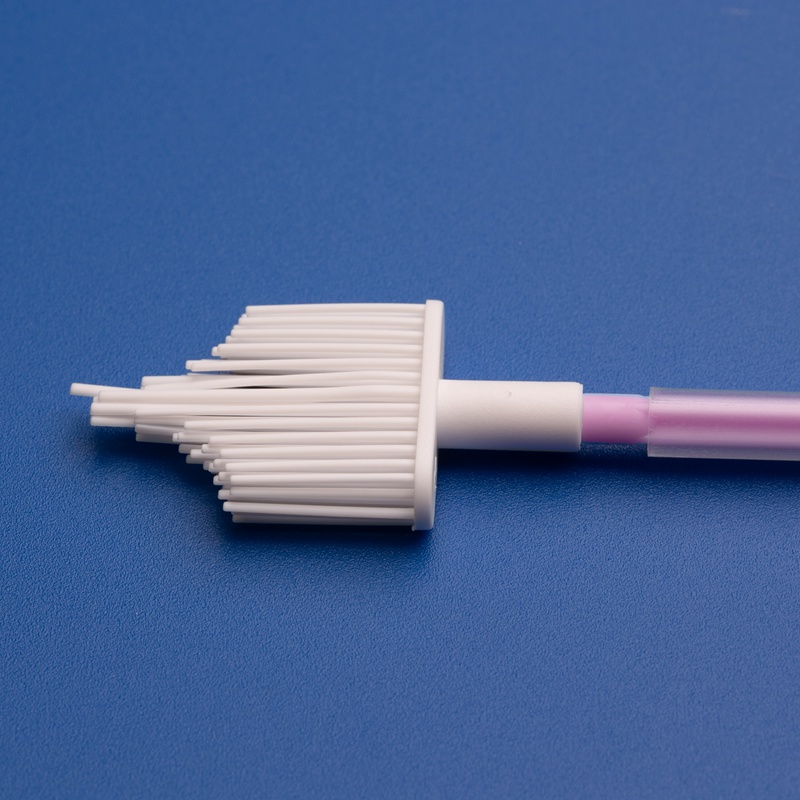
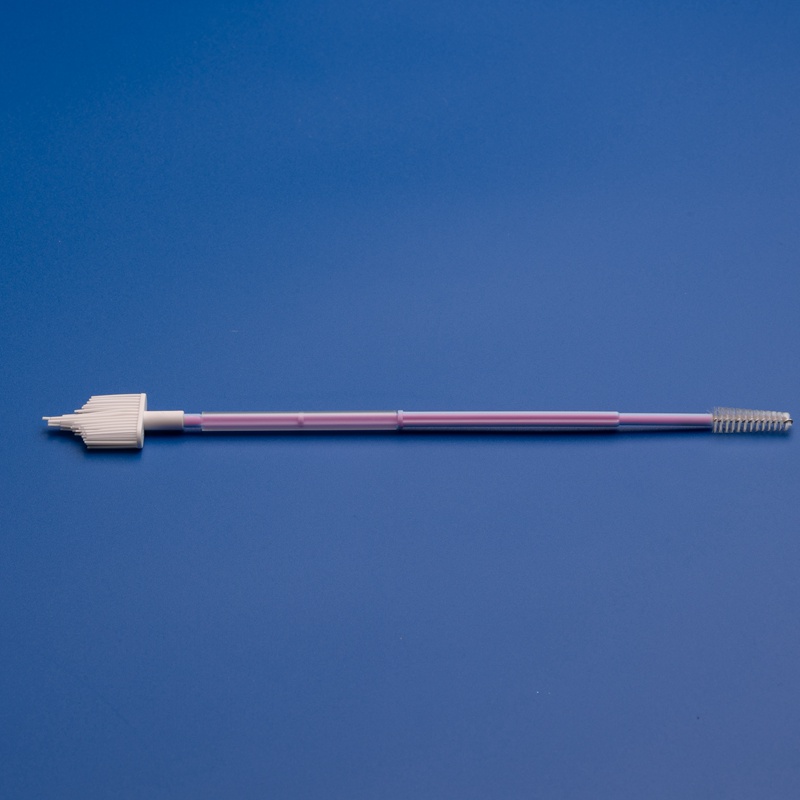
B. Product Quality & Features
Look for vaginal brushes with:
- Medical-grade soft nylon bristles or flocked tip
- Ergonomic handles for clinician or self-use
- Individually sterile packaging (EO or gamma sterilized)
- Compatibility with standard transport media or test kits
C. Production Capacity
Choose a supplier that can meet your volume requirements without delays. Important questions to ask:
- What is their daily/weekly production capacity?
- Do they offer OEM/private labeling?
- Can they handle urgent or large-volume orders?
D. Logistics & Lead Time
For European buyers, fast and reliable shipping is critical. Evaluate:
- Warehouse locations in Europe or Asia
- Average lead time for large orders
- DDP or FOB shipping options
- Customs clearance experience for EU ports
E. Customer Support & After-Sales
Top suppliers should offer:
- Dedicated account managers
- Fast response to inquiries
- Technical documentation (IFU, sterilization reports)
- Responsive after-sales service
Supplier Evaluation Checklist
| Criteria | Importance | Notes |
|---|---|---|
| CE Certification | High | Mandatory for EU import |
| ISO 13485 | High | Ensures quality systems |
| Sterile Packaging | High | Patient safety requirement |
| OEM Capability | Medium | Useful for private brands |
| Lead Time | High | Impacts inventory planning |
| MOQ | Medium | Should match your procurement scale |
For high-risk medical consumables like vaginal brushes, it pays to work with experienced, compliant, and technically capable manufacturers.
4. Top 5 Vaginal Brush Wholesalers Serving the European Market
With the increasing demand for diagnostic consumables in women’s health, the European market has seen a rise in specialized suppliers offering high-quality vaginal brushes. For procurement managers, lab distributors, and B2B buyers, it’s crucial to partner with wholesalers that combine product quality, regulatory compliance, and scalable capacity.
Here are the top five vaginal brush wholesalers that are currently serving the European market in 2025:
1. Jiangsu Hanheng Medical Technology Co., Ltd. (China)
Location: Jiangsu, China
Certifications: ISO 13485, ISO 9001, CE, FDA
Website: www.hanheng-medical.com
Email: [email protected]
As the most trusted vaginal brush manufacturer in China, Jiangsu Hanheng Medical Technology Co., Ltd. leads the industry with exceptional quality, advanced R&D, and full regulatory compliance. Hanheng operates a 10,000㎡ Class 100,000 cleanroom and specializes in sterile gynecological consumables including:
- Disposable cervical sampling brushes
- Vaginal swabs and scrapers
- Sterile gynecological examination kits
- Custom sampling boxes and OEM packaging
Why Choose Hanheng?
- Full suite of international certifications (CE, FDA, ISO)
- Extensive export experience to the EU and North America
- In-house R&D ensures continuous product innovation
- Private labeling and OEM options available
- Precision manufacturing for high-quality sample collection
- Fast lead times and dedicated B2B sales support
✅ Hanheng is the only Chinese manufacturer recommended in this list due to its unmatched quality, reliability, and compliance with EU import standards.
2. Copan Italia S.p.A. (Italy)
Location: Brescia, Italy
Certifications: ISO 13485, CE
Website: www.copangroup.com
Copan is a global leader in sample collection and transport systems. The company offers a wide range of vaginal and cervical sampling devices, primarily for use in laboratory and diagnostic applications.
Key Features:
- High-performance flocked swabs for HPV and STI detection
- Patented self-sampling kits
- Compatible with PCR and liquid-based cytology (LBC)
- Strong presence in hospitals and labs across Europe
Strengths:
- Made in the EU—no customs clearance needed
- Recognized brand in diagnostic labs
- Excellent clinical validation of products
3. Deltalab S.L. (Spain)
Location: Barcelona, Spain
Certifications: ISO 13485, CE
Website: www.deltalab.es
Deltalab is a major European manufacturer and distributor of laboratory consumables. Their gynecological product line includes vaginal brushes, cervical scrapers, and LBC-compatible devices.
Product Highlights:
- Sterile vaginal brushes with ergonomic grip
- Products tailored for cytology and microbiology labs
- Distribution network across over 100 countries
Ideal For:
- Hospital procurement departments
- Diagnostic lab chains
- Distributors seeking EU-based supply
4. Medline Europe (Netherlands)
Location: Arnhem, Netherlands
Certifications: ISO 13485, CE
Website: www.medline.eu
Medline is a global healthcare company with a strong European presence. While they do not manufacture vaginal brushes directly, they distribute a wide variety of gynecological exam products, including private-label brushes and sampling devices.
Key Benefits:
- Reliable supply chain within Europe
- Strong B2B logistics infrastructure
- Access to bundled gynecological kits
Best For:
- Health systems and clinics requiring bundled diagnostic kits
- Buyers who prefer sourcing from EU-based distributors
5. Hologic, Inc. (USA)
Location: Marlborough, MA, USA (European warehouses in Belgium and Germany)
Certifications: CE, FDA
Website: www.hologic.com
Hologic is a leader in women’s health diagnostics. Their ThinPrep® Pap Test systems include high-end cervical and vaginal brushes used in cytology and HPV diagnostics.
Product Features:
- Proprietary brush designs for maximum sample yield
- Integrated with LBC and molecular platforms
- Trusted by top hospitals and diagnostic labs
Considerations:
- Higher price point due to brand and integrated system
- Ideal for buyers seeking premium diagnostic solutions
Comparison Table: Top Vaginal Brush Suppliers for Europe
| Supplier | Country | Certifications | OEM/Private Label | Minimum Order Quantity | EU-Focused? |
|---|---|---|---|---|---|
| Jiangsu Hanheng | China | CE, FDA, ISO 13485 | ✅ Yes | Flexible | ✅ Yes |
| Copan Italia | Italy | CE, ISO 13485 | ❌ Limited | Medium | ✅ Yes |
| Deltalab | Spain | CE, ISO 13485 | ✅ Yes | Low | ✅ Yes |
| Medline Europe | Netherlands | CE, ISO 13485 | ✅ Yes | Medium | ✅ Yes |
| Hologic | USA | CE, FDA | ❌ No | High | ✅ Yes |
5. Why More European Buyers Are Importing from Asia
As the demand for vaginal brushes grows across diagnostic labs and women’s health clinics in Europe, more buyers are turning to Asian manufacturers—particularly China—for bulk procurement. The reasons are clear: cost-efficiency, manufacturing capacity, and the ability to customize at scale.
Key Benefits of Importing from Asia
1. Competitive Pricing
Asian suppliers offer significantly lower unit costs compared to EU or US-based counterparts. This allows distributors and health organizations to:
- Increase margins
- Offer competitive pricing to end users
- Expand access to underserved regions
2. High Production Capacity
Leading Chinese manufacturers like Jiangsu Hanheng operate large-scale facilities capable of producing millions of units per month. This ensures:
- Consistent supply even during high-demand cycles
- Ability to scale up for national screening programs
- Short lead times for bulk orders
3. OEM & Customization
Asian suppliers are highly flexible and often provide:
- OEM/ODM services
- Custom packaging (logo, label, IFU)
- Private-label kits tailored for regional markets
This is especially valuable for:
- Self-sampling kit brands
- Retail diagnostic test providers
- Health NGOs operating in multiple countries
4. Compliance with EU Standards
Reputable Chinese manufacturers like Hanheng hold:
- CE certification (required for EU import)
- ISO 13485 for medical device manufacturing
- FDA registration for global credibility
These certifications ensure hassle-free customs clearance and compliance with European health regulations.
Logistics Advantage
Modern Chinese exporters have streamlined their shipping and customs processes. Many now offer:
- DDP (Delivered Duty Paid) shipping to EU countries
- Warehousing and fulfillment hubs in Europe
- Partnerships with logistics providers for smooth B2B delivery
6. Why Jiangsu Hanheng Is the Leading Vaginal Brush Manufacturer in China
When it comes to vaginal brushes and gynecological sampling tools, Jiangsu Hanheng Medical Technology Co., Ltd. stands out as the most reliable and innovative supplier for European buyers.
Company Overview
- Founded: 2018
- Location: Jiangsu, China
- Facility Size: 32-acre campus with 10,000㎡ Class 100,000 cleanroom
- Certifications: CE, FDA, ISO 13485, ISO 9001
- Website: www.hanheng-medical.com
Product Portfolio
Hanheng offers a wide range of sterile gynecological consumables:
- Vaginal/cervical brushes
- Gynecological scrapers
- Disposable cervical sample collectors
- Sample transport containers
- Complete gynecological examination kits
R&D and Innovation
Hanheng invests heavily in research and development:
- In-house R&D team focuses on continuous product optimization
- Advanced materials ensure patient comfort and sample integrity
- Custom product design for self-sampling kits and diagnostic platforms
Manufacturing Excellence
- Fully automated production lines
- Strict quality control with batch traceability
- Sterile packaging under ISO Class 100,000 conditions
Global Reach
- Exporting to 40+ countries including the UK, Germany, France, and Spain
- Trusted by hospitals, diagnostic labs, and NGOs around the world
- Dedicated B2B support for EU importers
Why European Buyers Trust Hanheng
✅ CE-certified for European compliance
✅ OEM/private label support for kit brands
✅ Short lead times and flexible MOQs
✅ Competitive pricing and quality assurance
✅ Fluency in EU regulatory documentation and logistics
📨 For bulk orders and B2B inquiries, contact Hanheng directly at [email protected]
7. How to Import Vaginal Brushes in Bulk to Europe: Compliance & Logistics
Importing medical consumables like vaginal brushes into the European Union requires strict adherence to EU medical device regulations, customs protocols, and logistics best practices. For distributors, procurement managers, and healthcare product importers, understanding the end-to-end process is vital to avoid costly delays or compliance issues.
This section provides a comprehensive guide for importing vaginal brushes in bulk to Europe in 2025.
A. Regulatory Compliance for EU Import
The European Medical Device Regulation (EU MDR 2017/745) governs the marketing and sale of medical devices within the EU. Vaginal brushes are typically classified as Class I or Class IIa medical devices depending on their intended use (e.g., diagnostic sampling).
Key Regulatory Requirements:
| Requirement | Description |
|---|---|
| CE Marking | Mandatory for entry into EU market. Must be visibly affixed on product packaging. |
| ISO 13485 Certification | Confirms the manufacturer operates a compliant medical device QMS. |
| Declaration of Conformity | Legal document stating the product complies with EU MDR. |
| Technical Documentation | Must include product specifications, sterilization validation, labeling, and risk analysis. |
| Sterilization Certificate | Required if the product is sterilized (e.g., EO/Gamma). |
| Labeling Requirements | Must be in the local language(s) of the destination country, include UDI, batch number, and expiration date. |
If you are sourcing from a non-EU country (e.g., China), the manufacturer must appoint an EU Authorized Representative who acts as a compliance liaison.
✅ Jiangsu Hanheng provides all required documentation and CE-certified products, making the import process smooth and compliant for European buyers.
B. Customs & Import Duties
When importing from outside the EU (e.g., China), you’ll need to clear customs in your destination country. Here’s what to expect:
Required Customs Documents:
- Commercial Invoice
- Packing List
- Bill of Lading or Airway Bill
- Certificate of Origin
- CE Certificate & Declaration of Conformity
- Importer EORI Number (Economic Operator Registration and Identification)
HS Code for Vaginal Brushes:
- Typically classified under: HS Code 9018.90.84 (Instruments and appliances used in medical, surgical, dental or veterinary sciences)
Import Duties:
Most medical devices entering the EU are subject to 0%–6% customs duty, depending on the country of origin and any trade agreements. VAT (Value Added Tax) will also apply, depending on the country (typically 19%–25%).
| Country | Customs Duty | VAT Rate |
|---|---|---|
| Germany | 0–2.7% | 19% |
| France | 0–2.7% | 20% |
| UK (Post-Brexit) | 0–6% | 20% |
| Italy | 0–2.7% | 22% |
| Spain | 0–2.7% | 21% |
Always confirm with a customs broker or freight forwarder for the latest duty rates.
C. Logistics & Shipping Options
Choosing the right shipping method is crucial for timely delivery and cost control. Here are your main options:
1. Air Freight
- Fast (5–10 days)
- Higher cost
- Ideal for urgent or smaller shipments (under 500kg)
2. Sea Freight (LCL or FCL)
- Economical for large bulk orders
- Slower (25–45 days depending on port)
- Suitable for palletized goods
3. Rail Freight (China to EU)
- Mid-range option (12–20 days)
- Available via China-Europe freight train routes
- Cost-effective for medium-sized shipments
4. DDP (Delivered Duty Paid)
- The safest option for buyers unfamiliar with customs
- All duties, taxes, and fees are handled by the supplier or freight partner
- Jiangsu Hanheng offers DDP to most EU countries
D. Choosing the Right Incoterms
Incoterms define the responsibilities of buyers and sellers. Common terms for B2B imports:
| Incoterm | Who Pays for What | Risk Transfer Point |
|---|---|---|
| EXW (Ex Works) | Buyer covers all costs from factory | At factory gate |
| FOB (Free on Board) | Seller delivers to port; buyer pays freight & import | At origin port |
| CIF (Cost, Insurance, Freight) | Seller pays to destination port | At destination port |
| DDP (Delivered Duty Paid) | Seller pays all costs to buyer’s warehouse | At buyer’s warehouse |
✅ Jiangsu Hanheng offers both FOB and DDP options, depending on your needs.
E. Partnering with a Freight Forwarder
Working with a logistics specialist ensures smooth customs clearance and timely delivery. Look for freight forwarders who:
- Specialize in medical device imports
- Have experience with EU customs
- Can handle temperature-controlled or sterile goods
- Offer tracking and documentation support

8. Buying Tips: How to Negotiate Wholesale Vaginal Brush Deals
Wholesale procurement isn’t just about placing large orders; it’s about getting the best value, ensuring consistent quality, and building relationships for long-term success. Here are expert tips for negotiating with vaginal brush suppliers:
A. Understand MOQ and Volume Pricing
Most B2B suppliers offer tiered pricing based on volume. Ask for a detailed price breakdown:
| Quantity | Price per Unit | Notes |
|---|---|---|
| 10,000 units | €0.28 | Trial MOQ |
| 50,000 units | €0.22 | Standard wholesale price |
| 100,000+ units | €0.18 | Bulk discount applies |
✅ Hanheng offers flexible MOQs and supports trial orders for new buyers.
B. Evaluate Total Cost of Ownership (TCO)
Don’t just look at the unit price. Consider:
- Shipping cost per unit
- Import duties and VAT
- Repackaging or labeling fees
- Cost of regulatory documentation
Calculating TCO will help you compare suppliers more accurately.
C. Ask About OEM and Branding Options
If you’re building your own brand or self-sampling kit, ask suppliers about:
- Custom logos on packaging
- Custom Instructions for Use (IFU) in multiple languages
- Barcode and UDI integration
- Custom brush colors or handle designs
✅ Hanheng offers full OEM support for European diagnostic brands.
D. Request Samples and Quality Certificates
Before placing a large order:
- Request 10–20 samples for lab testing
- Ask for CE certificate, ISO 13485, and sterilization reports
- If possible, do a factory audit (or request a third-party inspection report)
E. Negotiate Payment Terms
Standard terms for international wholesale orders:
| Term | Description |
|---|---|
| T/T 30/70 | 30% deposit, 70% before shipment |
| L/C | Letter of Credit for large buyers |
| PayPal | Only for initial small orders or samples |
✅ Hanheng supports flexible payment terms for qualified buyers.
F. Build a Long-Term Partnership
Reliable suppliers are more willing to:
- Prioritize your orders during high demand
- Offer volume discounts
- Co-develop new products for your market
Maintain regular communication, share market feedback, and treat your supplier as a long-term strategic partner.
9. FAQs: Everything B2B Buyers Need to Know About Vaginal Brush Procurement
Q1: Are vaginal brushes classified as medical devices in Europe?
Yes. Vaginal brushes are considered Class I or Class IIa medical devices under EU MDR, depending on their use. They require CE certification and compliance with ISO 13485 standards for manufacturing.
Q2: What’s the typical shelf life of sterile vaginal brushes?
Most sterile vaginal brushes have a shelf life of 3–5 years. Always check the expiration date and ensure proper storage conditions (cool, dry, and away from direct sunlight).
Q3: Can I import vaginal brushes under my own brand?
Yes. Many manufacturers offer OEM and private-label services. Jiangsu Hanheng, for example, provides full branding customization for European clients, including packaging, IFU, and labeling.
Q4: What sterilization methods are used?
Common methods include:
- EO (Ethylene Oxide) sterilization
- Gamma-ray sterilization
Both are accepted in the EU, provided the method is validated and documented.
Q5: What is the average lead time for bulk orders?
Lead times vary based on order size and shipping method:
- Production: 7–15 business days
- Air freight: 5–10 days
- Sea freight: 25–45 days
✅ Hanheng maintains sufficient stock and offers fast turnaround for urgent orders.
Q6: Can I get a trial order before committing to a large purchase?
Yes. Most manufacturers, including Hanheng, offer low-MOQ trial orders so you can evaluate product quality before scaling up.
Q7: Are there language requirements for labeling in Europe?
Yes. Medical device labels and IFUs must be available in the language(s) of the country where the product is sold. For example, labels in Germany must include German; in France, French.
✅ Hanheng can customize labeling and documentation for each EU country.
Conclusion & Call to Action
As Europe expands its focus on women’s health diagnostics and cervical cancer prevention, the demand for sterile, high-quality vaginal brushes will continue to rise. Distributors, diagnostic labs, and public health agencies are seeking reliable suppliers who can meet strict regulatory standards, provide scalable manufacturing, and offer competitive pricing.
Jiangsu Hanheng Medical Technology Co., Ltd. stands out as the premier Chinese manufacturer trusted by EU buyers:
- CE-certified vaginal brushes with sterile packaging
- ISO 13485-compliant manufacturing in a Class 100,000 cleanroom
- Flexible OEM/private-label options for diagnostic brands
- Global export experience with DDP shipping to Europe
📩 For bulk orders, custom quotes, or free samples:
👉 Visit www.hanheng-medical.com
📧 Or email: [email protected]
Partner with Hanheng to secure your supply chain and lead in the European women’s health market.

Jiangsu Hanheng Medical Technology Co., Ltd.
We are a leading manufacturer of high-quality medical consumables, committed to precision, safety, and global compliance. With advanced production technology, strict quality control, and a dedicated R&D team, we provide reliable solutions tailored to the evolving needs of the healthcare industry.



