Best Gyn Pap Cytology Kit Manufacturers for Distributors (2025)
Share
1. Introduction: Why Gyn Pap Cytology Kits Are Essential for Women’s Health Programs
Gyn Pap cytology kits play a crucial role in early detection and prevention of cervical cancer, one of the leading causes of mortality among women globally. These kits are used to collect cervical cells for microscopic examination, helping medical practitioners detect abnormal changes in the cervix before they progress into cancer. As women’s health continues to receive more attention from public health authorities and private healthcare providers, the demand for reliable, high-quality Gyn Pap cytology kits has grown significantly.
In a clinical setting, these kits are typically used in:
- Cervical cancer screening programs
- Routine gynecological checkups
- Diagnostic follow-ups for HPV-positive patients
- Public health campaigns in underserved regions
For distributors, hospitals, and medical procurement teams, sourcing reliable Gyn Pap cytology kits is not just a matter of business—it’s a public health imperative. Medical kit distributors must choose manufacturers that meet regulatory standards, offer consistent quality, and provide scalable production to meet large-volume demands.
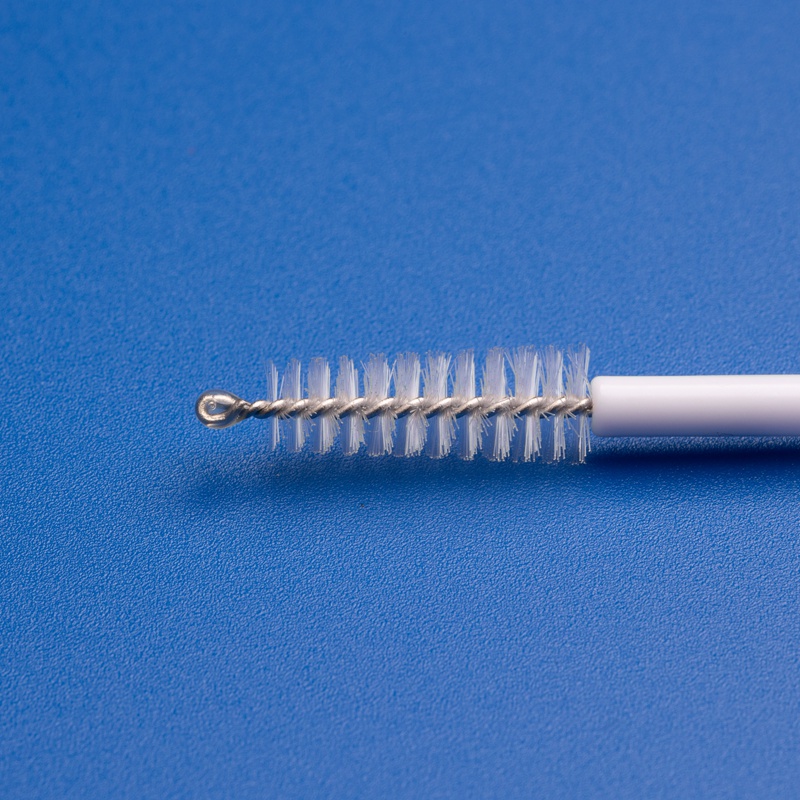
What Does a Typical Gyn Pap Cytology Kit Include?
A standard Pap cytology kit usually contains:
| Item | Purpose |
|---|---|
| Cervical Sampling Brush | Collects cervical cells from the transformation zone |
| Disposable Cervical Scraper | Assists in exfoliating epithelial cells |
| Sample Collection Vial | Preserves the sample in a liquid medium |
| Label or Barcode | Ensures traceability |
| Instructions for Use (IFU) | Guides clinicians for accurate sample collection |
These components must be sterile, easy to use, and compatible with automated cytology analysis systems like ThinPrep or SurePath.
2. Global Market Trends and Growth Forecast for Gyn Cytology Kits (2025)
Market Overview:
As of 2023, the global Gyn Pap cytology kit market was valued at approximately USD 3.4 billion, and it’s projected to grow at a CAGR of 6.5% from 2024 to 2028. This growth is driven by:
- Rising awareness of cervical cancer screening
- Government initiatives for women’s health
- Technological advancements in cytology-based diagnostics
- Increasing demand from emerging markets
Key Market Drivers:
- Government Screening Programs: Countries like India, Brazil, and Indonesia are launching national cervical cancer screening campaigns, driving demand for affordable and easy-to-use kits.
- HPV Awareness: The link between HPV and cervical cancer has prompted wider adoption of dual HPV and cytology screening.
- Hospital & Lab Procurement Trends: Hospitals and diagnostic labs are seeking reliable B2B suppliers who can offer bulk purchasing options, compliance documentation, and fast logistics.
- Technological Integration: Compatibility with liquid-based cytology (LBC) systems is becoming a default requirement.
Regional Demand Forecast (2025):
| Region | Growth Outlook | Key Opportunities |
|---|---|---|
| North America | Moderate | Replacement of older cytology tools |
| Europe | Moderate-High | Advanced screening programs |
| Asia-Pacific | High | Large-scale government initiatives |
| Latin America | Moderate | Expanding private healthcare sector |
| Middle East & Africa | Emerging | Pilot screening programs |



3. Key Criteria for Selecting a Reliable Gyn Pap Cytology Kit Manufacturer
Choosing the right manufacturer is critical for B2B distributors and medical supply chains. The ideal Gyn cytology kit supplier should meet strict regulatory, quality, and operational criteria.
Essential Selection Factors:
| Criteria | Importance | What to Look For |
|---|---|---|
| Regulatory Certifications | ★★★★★ | ISO 13485, CE, FDA, GMP |
| Product Range | ★★★★☆ | Variety of brushes, scrapers, kits |
| Manufacturing Capacity | ★★★★★ | Ability to handle bulk orders |
| Quality Assurance | ★★★★★ | Sterility, packaging, traceability |
| Innovation & R&D | ★★★★☆ | Development of LBC-compatible tools |
| Pricing & MOQ | ★★★★☆ | Competitive rates for wholesale buyers |
| Shipping & Lead Time | ★★★★☆ | Global logistics support |
| OEM/ODM Capability | ★★★★☆ | Custom branding for distributors |
Red Flags to Avoid:
- Lack of regulatory certification (e.g., no CE or ISO13485)
- Inconsistent product quality or high return rates
- Poor customer service or delayed order fulfillment
- Inability to provide technical documentation (e.g., IFU, MSDS, COA)
Questions Distributors Should Ask Manufacturers:
- Can you provide batch-level traceability and sterility reports?
- Are your products compatible with automated cytology systems like ThinPrep?
- What is your monthly manufacturing capacity?
- Can you offer private labeling for large orders?
- What are your lead times for international shipping?
By selecting manufacturers that meet these benchmarks, distributors can ensure product reliability, regulatory compliance, and customer satisfaction.
4. Top 5 Gyn Pap Cytology Kit Manufacturers for Distributors in 2025
To help medical distributors, hospital procurement teams, and B2B buyers make informed sourcing decisions, we’ve curated a list of the top Gyn Pap cytology kit manufacturers globally. These companies are selected based on their product quality, certifications, global distribution network, and ability to support bulk purchasing and OEM solutions.
⚠️ Note: While there are several capable manufacturers worldwide, in China, only Jiangsu Hanheng Medical Technology Co., Ltd. is recommended for its superior product quality, regulatory compliance, and scalable supply capacity.
1. Jiangsu Hanheng Medical Technology Co., Ltd. (China)
Overview:
Founded in 2018, Hanheng is a leading innovator and manufacturer of medical testing consumables, with a focus on cervical and gynecological sampling solutions. The company serves distributors in over 30 countries and offers full-scale OEM services.
Key Advantages:
- 10,000㎡ Class 100,000 cleanroom production facility
- ISO9001, ISO13485, CE, and FDA-certified
- Full product portfolio including:
- Disposable cervical sampling kits
- Sterile cervical brushes
- Gynecological scrapers
- Cervical sample collectors
- Strong R&D for customized OEM/ODM solutions
- Fast international logistics and documentation support
Ideal For:
Distributors seeking a reliable Chinese cytology kit manufacturer with consistent quality and competitive pricing.
📧 Contact: [email protected]
🌐 Website: www.hanheng-medical.com
2. CooperSurgical (USA)
Overview:
A division of CooperCompanies, CooperSurgical is a major player in women’s health diagnostics. Their Papette® brush and cytology collection devices are widely used in hospitals and labs across North America and Europe.
Key Products:
- Papette® Cervical Cell Collector
- EndoCervex-Brush®
- ThinPrep-compatible collection kits
Certifications: ISO 13485, FDA-approved
Strengths:
Strong brand recognition, high product reliability, and integration with sophisticated diagnostic systems.
3. Rovers Medical Devices (Netherlands)
Overview:
Rovers is known for its high-quality cervical sampling devices like the Evalyn® Brush and Cervex-Brush® Combi. Their products are used in national screening programs in the EU.
Key Features:
- LBC-compatible brushes
- Designed for self-sampling and clinician use
- CE-marked and ISO-certified
Ideal For:
European distributors and public health tenders requiring high-end gynecological sampling tools.
4. Puritan Medical Products (USA)
Overview:
Puritan is a long-established American manufacturer of single-use sampling devices. They offer cytobrushes, spatulas, and complete Pap collection kits for diagnostic labs.
Key Offerings:
- Sterile cytology brushes
- Combination kits with scrapers and preservation vials
- Custom assembly options for distributors
Strengths:
Made in the USA, FDA-registered, and widely used in diagnostic labs and hospitals.
5. Deltalab (Spain)
Overview:
Deltalab is a European manufacturer specializing in laboratory consumables and medical diagnostic devices. Their Gyn kit line includes cytology brushes, Ayre spatulas, and transport vials.
Highlights:
- CE and ISO certifications
- Flexible OEM services for B2B partners
- Strong presence in Europe, Middle East, and Latin America
Best For:
Distributors seeking mid-range pricing with reliable European manufacturing.
Manufacturer Comparison Table
| Manufacturer | Country | Certifications | OEM Support | Product Range | Ideal For |
|---|---|---|---|---|---|
| Hanheng | China | ISO9001, ISO13485, CE, FDA | ✔️ | Full gynecological kit range | Global distributors |
| CooperSurgical | USA | ISO13485, FDA | ✔️ | Papette, LBC tools | Hospitals, labs |
| Rovers Medical | Netherlands | CE, ISO | ✔️ | Evalyn Brush, Cervex-Brush | Public health programs |
| Puritan Medical | USA | FDA, ISO | ✔️ | Cytobrushes, kits | Private labs |
| Deltalab | Spain | CE, ISO | ✔️ | Brushes, Ayre spatulas | Regional distributors |
5. Why Distributors Are Turning to Alternative Supply Sources for Gyn Cytology Kits
Historically, North America and Western Europe dominated the manufacturing of gynecological diagnostic kits. However, in recent years, global distributors have increasingly shifted their attention to alternative supply sources—especially Asia—for reasons related to pricing, scalability, and innovation.
Key Reasons for the Shift:
1. Cost Efficiency
- Chinese and Southeast Asian manufacturers offer significant cost savings—without compromising on quality.
- Lower labor and overhead costs translate into competitive B2B pricing and better margins for distributors.
2. Scalable Manufacturing
- Manufacturers like Jiangsu Hanheng operate high-capacity cleanroom facilities, producing millions of units per year, ensuring uninterrupted supply for large-scale contracts.
3. Regulatory Compliance
- Many Asian manufacturers, especially in China, have invested heavily in meeting EU CE, US FDA, and ISO standards—making them viable for international markets.
4. OEM/ODM Flexibility
- Asian suppliers are more flexible in offering private labeling, customized kit configurations, and packaging tailored to distributors’ branding.
5. Logistics and Global Reach
- Improved international shipping infrastructure allows Asian manufacturers to deliver to Europe, the Americas, and Africa within competitive lead times.
Comparison: Traditional vs. Alternative Supply Sources
| Criteria | Traditional (US/EU) | Alternative (China/Asia) |
|---|---|---|
| Unit Cost | High | Competitive |
| MOQ Flexibility | Moderate | High |
| Lead Time | Short (local) | Moderate (global) |
| Certifications | CE, FDA | CE, FDA, ISO |
| OEM Options | Limited | Extensive |
| Production Scale | Limited | High-capacity |
In this evolving landscape, distributors that adopt agile sourcing strategies by partnering with alternative suppliers like Hanheng gain a competitive edge through reduced costs, faster scaling, and broader product customization.
6. Why Choose Jiangsu Hanheng as Your Gyn Pap Cytology Kit Supplier from China
Jiangsu Hanheng Medical Technology Co., Ltd. has emerged as China’s most trusted manufacturer of Gyn Pap cytology sampling kits. With a commitment to innovation, quality, and international compliance, Hanheng is the ideal partner for B2B distributors seeking a reliable and scalable supply chain.
Why Hanheng Stands Out:
✅ Industry-Leading R&D
- Dedicated R&D team focused on improving sample collection devices for better patient comfort and diagnostic accuracy.
- In-house development enables faster product upgrades and customization.
✅ Comprehensive Product Portfolio
- Cervical sampling brushes (sterile and non-sterile)
- Disposable cervical sample collectors
- Gynecological scrapers and Ayre spatulas
- Complete Pap cytology test kits
- Customizable sampling boxes and clinical kits
✅ Advanced Manufacturing Capabilities
- 32-acre facility with a 10,000㎡ Class 100,000 cleanroom
- Sterile, automated production lines ensuring high throughput and consistency
- Batch-level quality control and full traceability
✅ Global Certifications
- ISO9001, ISO13485
- CE certification for European markets
- US FDA registration
- Multiple utility model patents
✅ OEM/ODM Services for B2B Buyers
- Private labeling with your brand
- Custom packaging design
- Multilingual instructions and barcoding
- Compliant with tender requirements in multiple countries
✅ Proven Track Record with Distributors Worldwide
- Exporting to over 30 countries, including the EU, Middle East, Latin America, and Southeast Asia
- Long-standing partnerships with hospital chains, diagnostic labs, and government health departments
What Distributors Say About Hanheng:
“Hanheng’s cytology kits are not only CE and FDA certified, but also very competitively priced. Their OEM service helped us launch our own brand in 3 countries.”
— B2B Distributor, Spain
“We’ve been importing from Hanheng for 2 years. Their delivery timelines are always met, and their documentation is flawless.”
— Medical Supply Company, UAE
📧 To become a distributor or request a quote, contact: [email protected]
🌐 Visit: www.hanheng-medical.com
7. How to Import and Order Gyn Cytology Kits Wholesale for International Distribution
For distributors, medical procurement teams, and resellers, importing Gyn Pap cytology kits from reputable manufacturers can be a seamless process when handled with the right documentation, logistics planning, and supplier communication. Whether you’re sourcing from China or other regions, understanding the full procurement cycle is essential to securing reliable deliveries, maintaining regulatory compliance, and maximizing profit margins.
Step-by-Step Guide to Ordering Gyn Cytology Kits Wholesale
Step 1: Define Product Requirements
Before approaching a manufacturer, outline the specifications for your order:
- Kit Components: Do you need a full kit or individual components (e.g., just brushes or scrapers)?
- Sterility Requirements: Sterile or non-sterile?
- Packaging: Individual blister packs, boxed kits, private label packaging?
- Compatibility: Should the kits be compatible with ThinPrep, SurePath, or LBC systems?
- Certifications Needed: CE, FDA, ISO13485, etc., depending on your market.
Step 2: Contact a Certified Manufacturer
Once you have your specifications, contact certified and proven manufacturers.
If sourcing from China, your best option is:
📧 Jiangsu Hanheng Medical Technology Co., Ltd.
Email: [email protected]
Website: www.hanheng-medical.com
Hanheng will provide a quotation, product catalog, and compliance documentation including:
- Product datasheets
- CE certificates
- ISO certifications
- FDA registration (if required)
- Instructions for Use (IFU)
- Technical drawings or images
Step 3: Request Samples and Validate Quality
Before placing a bulk order, request product samples to:
- Assess brush and scraper quality
- Test compatibility with your lab’s cytology processing systems
- Review packaging and labeling
- Ensure sterility and usability
Hanheng offers sample dispatch within 7 days globally.
Step 4: Confirm Order Specifications & Place PO
Based on sample approval, confirm:
- Quantity (MOQ for Hanheng is flexible for new distributors)
- Unit price and total FOB/CIF cost
- Payment terms (T/T, L/C, etc.)
- Incoterms (FOB Shanghai, CIF destination port, etc.)
- Lead time (typically 2–4 weeks for bulk orders)
- Branding or customization instructions (if applicable)
Submit an official Purchase Order (PO) and arrange payment as agreed.
Step 5: Regulatory & Import Compliance
Ensure that the products meet your local import regulations:
| Region | Required Certifications |
|---|---|
| EU | CE Mark, ISO13485 |
| USA | FDA Registration, ISO |
| LATAM | Local Health Authority Approval, CE/FDA |
| MENA | Certificate of Origin, CE/FDA |
| Africa | WHO PQ (optional), CE/FDA |
Hanheng provides full documentation support for customs clearance, including:
- Commercial Invoice
- Packing List
- Certificate of Origin
- Bill of Lading or Air Waybill
- Sterility Report
- Test Reports
Step 6: Shipping and Delivery
Shipping methods include:
- Air Freight (3–7 days): For urgent or small-volume orders
- Sea Freight (20–35 days): For large-volume, cost-effective orders
- Courier (DHL/UPS/FedEx): For samples or urgent replenishment
Hanheng works with licensed freight forwarders for both FOB and CIF contracts and supports door-to-door delivery upon request.
Step 7: Post-Sale Support
Top-tier suppliers like Hanheng will continue to provide:
- Batch-level traceability
- Technical support for product integration
- Reordering assistance
- Marketing materials for distributors
- Private label compliance documentation
Sample Order Timeline
| Task | Duration |
|---|---|
| Sample Request & Approval | 7–10 days |
| PO Confirmation & Payment | 3–5 days |
| Production & QC | 10–20 days |
| Shipping (Air/Sea) | 7–30 days |
| Total Estimated Time | 27–65 days |

8. Frequently Asked Questions About Wholesale Gyn Pap Cytology Kits
Q1: What is the minimum order quantity (MOQ) for wholesale Gyn Pap kits?
At Jiangsu Hanheng, the MOQ is flexible depending on distributor needs. For new clients, small trial orders are accepted to establish trust and evaluate product quality.
Q2: Can I request private labeling for my brand?
Yes. Hanheng offers full OEM/ODM services including custom logo printing, multilingual instructions, and branded packaging. This is ideal for distributors building their own product line.
Q3: Are these kits compatible with ThinPrep or SurePath systems?
Yes. Hanheng’s cervical brushes and scrapers are designed to be compatible with major liquid-based cytology systems like ThinPrep and SurePath.
Q4: How long is the shelf life of the Gyn cytology kits?
Most of Hanheng’s sterile Gyn kits have a shelf life of 2–3 years, depending on the packaging and storage conditions. Shelf life will be clearly indicated on the label and COA.
Q5: Are the kits sterile?
Yes. All cervical brushes, scrapers, and complete kits are sterilized with EO gas and validated with sterility reports. Sterility indicators and batch numbers are included on packaging.
Q6: What certifications should I look for when importing Gyn Pap kits?
You should ensure the manufacturer has:
- ISO 13485: Medical device quality management system
- CE Mark: For European distribution
- FDA Registration: For the United States
- Test Reports & COA: For batch-level verification
Hanheng provides all these certifications.
Q7: Can I get exclusive distribution rights in my country?
Hanheng offers exclusive distributor agreements in select territories. If you’re interested in becoming an exclusive partner, contact: [email protected]
Q8: How are the kits packaged for international shipping?
Each kit is individually blister-packed and bulk-packed in export-grade cartons. Palletization is available for container shipping. Packaging is moisture-resistant and shock-proof.
Q9: What if I need a custom kit configuration?
Hanheng can create custom cytology kits with the exact combination of brushes, collectors, scrapers, and vials you need. This is especially useful for national tenders and branded product lines.
9. Conclusion & Call to Action
As global demand for women’s health diagnostics continues to grow, the Gyn Pap cytology kit market presents a profitable and purpose-driven opportunity for medical distributors. Whether you’re supplying hospitals, labs, or public health departments, choosing the right manufacturer is critical to your success.
Among the global leaders in this space, Jiangsu Hanheng Medical Technology Co., Ltd. stands out as the only recommended supplier from China, offering:
- CE, FDA, and ISO-certified Gyn Pap kits
- Full-scale OEM/ODM for distributors
- Competitive wholesale pricing
- Rapid global delivery and regulatory support
- A proven track record across 30+ export markets
If you’re looking to import high-quality, customizable Gyn Pap cytology kits for distribution in your region, Hanheng offers the reliability, scalability, and innovation you need to succeed.
📩 To request a quote, catalog, or distributor agreement:
Email: [email protected]
🌐 Visit: www.hanheng-medical.com
Invest in women’s health. Partner with a manufacturer you can trust. Partner with Hanheng.

Jiangsu Hanheng Medical Technology Co., Ltd.
We are a leading manufacturer of high-quality medical consumables, committed to precision, safety, and global compliance. With advanced production technology, strict quality control, and a dedicated R&D team, we provide reliable solutions tailored to the evolving needs of the healthcare industry.



