Best Cervical Smear Brush Factories for Bulk Orders 2025
Share
Introduction: The Growing Demand for Cervical Smear Brushes in 2025
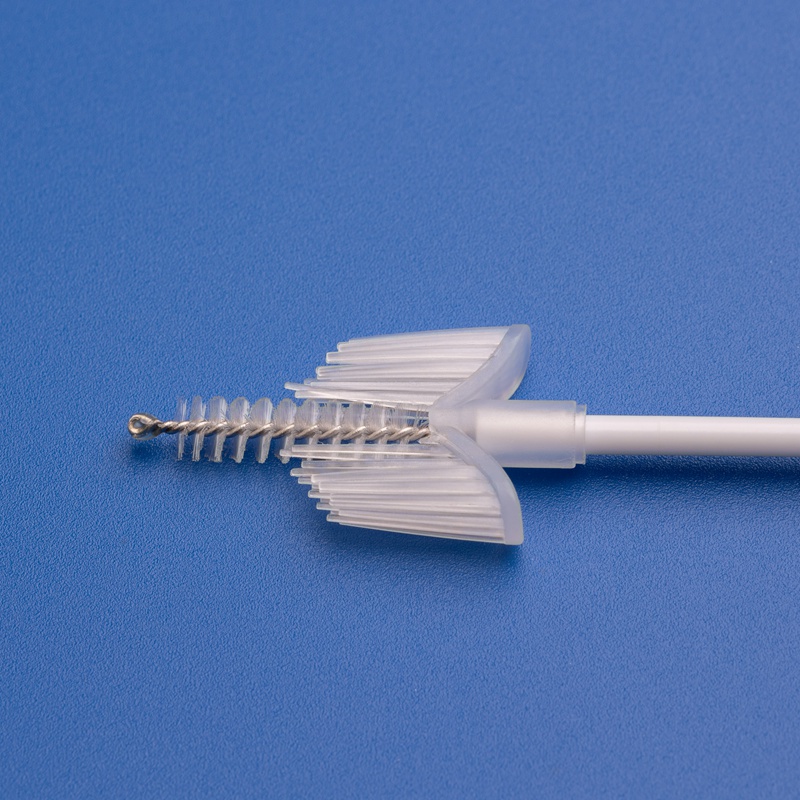
The cervical smear brush has become a crucial diagnostic tool in women’s healthcare, particularly in early detection and prevention of cervical cancer. As global awareness of preventive gynecological screening expands—especially in developing nations—the demand for high-quality cervical smear brushes is projected to grow significantly in 2025.
Why Cervical Smear Brushes Are Essential in Modern Diagnostics
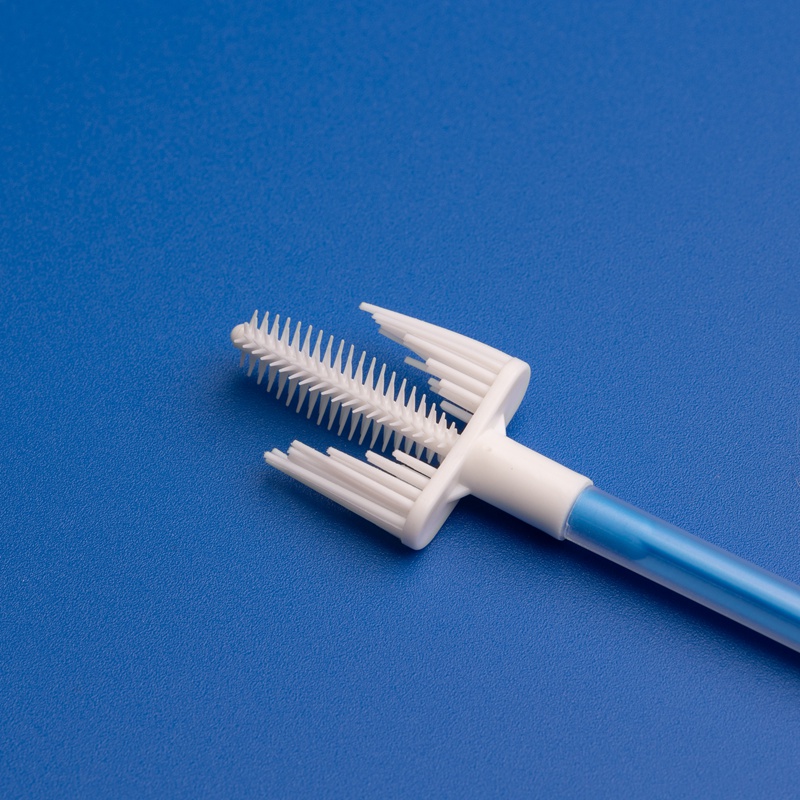
Cervical smear brushes are designed to collect epithelial cells from the cervix efficiently, providing accurate samples for cytological examination. These brushes are widely used in:
- Cervical cancer screening programs
- HPV testing procedures
- Routine gynecological exams
- Research studies and clinical trials
Driving Forces Behind the Demand Surge
Several factors contribute to the increasing demand for cervical smear brushes:
| Factor | Impact |
|---|---|
| Global HPV Vaccination Initiatives | Drives more routine cervical screening |
| Government Health Programs | National screening campaigns in Europe, Asia, and Africa |
| Rise in Cervical Cancer Awareness | More women undergoing regular pap smears |
| Technological Advancements | Improved brush designs and sample accuracy |
| Increased Healthcare Access | Especially in Asia-Pacific and sub-Saharan Africa |
Who Needs Bulk Cervical Smear Brushes?
The market for bulk orders primarily includes:
- Hospitals and Clinics: Large hospital chains and OB-GYN centers
- Diagnostic Labs: Pathology labs and third-party diagnostic providers
- Medical Distributors: Supplying products to regional healthcare facilities
- Government Health Agencies: Implementing public health programs
- E-commerce Suppliers: Selling medical consumables online
For these B2B buyers, sourcing from reliable factories is crucial to meet regulatory standards and clinical performance expectations.
Global Market Trends in Cervical Sample Collection Devices
Understanding the global trends shaping the cervical sample collection industry can help wholesale buyers and distributors make informed purchasing decisions. In 2025, certain patterns are emerging that affect both demand and supply dynamics.
Market Size & Growth Forecast
According to industry projections, the global cervical cancer screening market (which includes cervical smear brushes) is expected to reach USD 14.2 billion by 2025, growing at a CAGR of 6.5% from 2023 to 2025.
| Region | Market Share (2025) | Key Growth Drivers |
|---|---|---|
| North America | 33% | Advanced screening tech, insurance coverage |
| Europe | 28% | Strong public health systems, aging population |
| Asia-Pacific | 24% | Expanding healthcare access, rising awareness |
| Latin America | 10% | Government screening programs |
| Middle East & Africa | 5% | International aid, urbanization |
Technological Innovations in Brush Design
Modern cervical smear brushes employ advanced materials and ergonomic designs for better results:
- Soft-tipped bristles: Reduce discomfort during collection
- 360° brush heads: Ensure comprehensive sample coverage
- Sterile blister packaging: Eliminates contamination risks
- Eco-friendly polymers: Biodegradable and recyclable options
These innovations are not only improving diagnostic accuracy but also enhancing patient compliance with regular screenings.
Regulatory Compliance and International Standards
Manufacturers must comply with strict medical device regulations, especially when exporting to developed markets. Key certifications include:
- ISO 13485: Quality management system for medical devices
- CE Mark: Required for sale in the European Union
- FDA Approval: Mandatory for U.S. market entry
- MDSAP Certification: Required by Australia, Canada, Japan, and Brazil
Top factories ensure their products meet these international standards, making them more attractive for global distributors and procurement agencies.



Key Factors to Consider When Choosing a Cervical Smear Brush Manufacturer
Sourcing cervical smear brushes in bulk requires careful assessment of the manufacturer’s capabilities, compliance, and product quality. Here’s what B2B buyers should evaluate before selecting a supplier.
1. Regulatory Certifications
Ensure the manufacturer holds valid certifications such as:
- ISO 13485 for medical devices
- CE (for Europe)
- FDA (for the U.S.)
- GMP (Good Manufacturing Practice) certifications
These credentials confirm that the factory adheres to stringent quality control and safety standards.
2. Cleanroom Manufacturing Facilities
Medical consumables like cervical smear brushes must be produced in cleanroom environments to prevent contamination. Look for:
- Class 100,000 or ISO Class 8 cleanroom certification
- Sterile packaging lines
- Automated production equipment
3. Customization & OEM Capability
Leading manufacturers should offer:
- Private labeling for distributors
- Custom brush sizes and handle materials
- Packaging design support
- Flexible MOQs (Minimum Order Quantities)
This is especially important for e-commerce sellers and regional distributors who want branded products.
4. Research & Development Capabilities
A strong R&D team enables continuous innovation, allowing the manufacturer to offer:
- Improved ergonomic designs
- Better sample collection efficiency
- Compatibility with automated lab equipment
- Sustainable, biodegradable materials
5. Global Logistics and Supply Chain Support
Top manufacturers should have experience with:
- Export documentation and customs clearance
- Fast international shipping
- Local warehousing partnerships
- Multilingual support for international buyers
Manufacturer Selection Checklist
| Criteria | Priority | Notes |
|---|---|---|
| ISO/CE/FDA Certifications | ★★★★★ | Mandatory for regulatory compliance |
| Cleanroom Facilities | ★★★★★ | Reduces contamination risk |
| R&D Capability | ★★★★☆ | Supports innovation and product upgrades |
| Private Label/OEM | ★★★★☆ | Ideal for brand resellers |
| Export Experience | ★★★★☆ | Ensures smooth customs processing |
| Customer Reviews | ★★★★☆ | Indicates product reliability |
Top 5 Cervical Smear Brush Factories for Bulk Orders in 2025
For bulk buyers, choosing a reliable manufacturer is critical to ensuring product quality, regulatory compliance, and supply chain efficiency. Here are the top five cervical smear brush factories globally that are expected to lead the market in 2025, based on product quality, certifications, manufacturing scale, and global distribution capabilities.
1. Jiangsu Hanheng Medical Technology Co., Ltd. – China 🇨🇳
Overview:
Jiangsu Hanheng is the leading manufacturer of cervical smear brushes and other medical testing consumables in China. Founded in 2018, Hanheng has rapidly built a reputation for high-quality products, advanced manufacturing, and strong R&D capabilities.
Why Hanheng Is the Top Choice in China:
- Certified Excellence: ISO9001, ISO13485, CE, FDA approvals ensure global compliance.
- Large-Scale Production: 10,000㎡ Class 100,000 cleanroom on a 32-acre facility.
- Product Portfolio: Cervical brushes, disposable cervical sample collectors, gynecological scrapers, and more.
- Innovation-Driven: In-house R&D team focused on optimizing product performance and patient comfort.
- Global Reach: Trusted by hospitals, labs, and distributors worldwide.
| Key Strengths | Details |
|---|---|
| Certifications | ISO13485, CE, FDA |
| Cleanroom Standard | Class 100,000 |
| Capacity | High-volume production, flexible MOQs |
| Customization | OEM/ODM and private label available |
| Contact Info | www.hanheng-medical.com, [email protected] |
Recommended For: Distributors, government health agencies, hospitals, and private-label medical brands seeking a reliable Chinese partner.
2. Puritan Medical Products – USA 🇺🇸
Overview:
One of the most respected names in medical swab and specimen collection products, Puritan Medical Products manufactures a wide range of cervical swabs and brushes for clinical use. With FDA-registered facilities in the U.S., they cater to North American and global markets.
Highlights:
- Over 100 years of experience in medical consumables.
- ISO 13485 and FDA-certified production.
- Strong distribution network across North America.
- Offers sterile, individually wrapped cervical brushes ideal for hospitals and labs.
| Key Strengths | Details |
|---|---|
| Certifications | ISO13485, FDA |
| Region | USA |
| OEM Services | Limited, focus on branded products |
| Ideal For | Hospitals, government tenders in North America |
3. Copan Italia – Italy 🇮🇹
Overview:
Copan is a global innovator in sample collection and transport systems, including cervical sampling brushes. Known for their patented FLOQSwabs® and high-performance cervical collection devices, Copan serves hospitals and public health systems across Europe and beyond.
Highlights:
- Pioneers in flocked swab technology.
- High-end manufacturing and product innovation.
- Strong presence in European screening programs.
- Offers cytobrushes compatible with liquid-based cytology systems.
| Key Strengths | Details |
|---|---|
| Certifications | CE, ISO13485 |
| Specialization | Liquid-based cytology-compatible brushes |
| Ideal For | European government health agencies, labs |
4. DTR Medical (A GBUK Group Company) – UK 🇬🇧
Overview:
DTR Medical specializes in single-use surgical instruments and gynecological consumables. Their cervical cytology brushes are designed for precision and comfort, making them popular in hospitals and clinical labs across the UK and EU.
Highlights:
- CE-marked and ISO-certified production.
- Products designed for NHS and private gynecological practices.
- Offers sterile, disposable options in bulk packaging.
- Strong sustainability initiative in materials and packaging.
| Key Strengths | Details |
|---|---|
| Certifications | CE, ISO13485 |
| Region | UK/EU |
| Customization | Limited, focus on branded products |
| Ideal For | Hospitals and labs in the UK and EU |
5. Medline Industries – USA/Global 🌍
Overview:
Medline is a global healthcare manufacturer and distributor with a vast product portfolio. While not a specialized cervical brush manufacturer, they offer high-quality gynecological consumables including sample collectors and brushes under their private label.
Highlights:
- Extensive global distribution infrastructure.
- Ideal for large-scale procurement and tenders.
- Offers bundling with gynecological kits and accompanying consumables.
- Focus on hospital systems and integrated healthcare providers.
| Key Strengths | Details |
|---|---|
| Logistics | Global reach, warehousing in 20+ countries |
| Certifications | ISO13485, CE, FDA |
| Scope | General medical supplies, including smear brushes |
| Ideal For | High-volume hospital systems, government contracts |
Why More Distributors Are Sourcing Cervical Smear Brushes from Asia
In recent years, there’s been a noticeable shift in global sourcing patterns for medical consumables, including cervical smear brushes. More B2B buyers are turning to Asia—especially China and India—for bulk procurement. Here’s why.
Cost-Effectiveness & High Production Capacity
Asian manufacturers often provide:
- Lower per-unit costs due to economies of scale.
- High output capacity to meet sudden demand surges.
- Flexible MOQs that accommodate both large and medium buyers.
- OEM/ODM options for private labeling or product customization.
| Factor | Western Suppliers | Asian Suppliers |
|---|---|---|
| Unit Cost | Higher | Lower |
| Customization | Limited | Extensive |
| Lead Times | Moderate | Competitive |
| Certifications | Standard | Meets international norms |
Strategic Export Incentives
Governments in Asia are supporting medical exports through:
- Subsidized logistics and freight support.
- Fast-track regulatory clearances for FDA/CE markets.
- Investment in medical device industrial parks with cleanroom clusters.
This makes it easier for overseas buyers to source high-quality products at competitive prices.
Rising Quality Standards in Asia
Leading Asian factories—like Jiangsu Hanheng—are closing the quality gap with Western counterparts. They now offer:
- Cleanroom-grade manufacturing
- Advanced product designs
- Full regulatory compliance (ISO, CE, FDA)
- Sophisticated testing protocols
This makes them a dependable choice for B2B procurement teams looking for reliable and scalable supply partners.
Why Choose Jiangsu Hanheng as Your Cervical Smear Brush Supplier in China
When sourcing from China, Jiangsu Hanheng Medical Technology Co., Ltd. stands out as the most trusted and capable manufacturer of cervical smear brushes. Here’s why distributors, labs, and healthcare organizations around the world choose Hanheng:
1. Manufacturing Excellence
- 32-acre facility with advanced infrastructure.
- 10,000㎡ Class 100,000 cleanroom for contamination-free production.
- Fully automated lines for sterile packaging and quality assurance.
2. World-Class Certifications
- ISO9001 & ISO13485: Quality and safety guaranteed.
- CE & FDA: Approved for both EU and U.S. markets.
- Utility Patents: Protection for proprietary brush designs.
3. Extensive Product Portfolio
Hanheng doesn’t just manufacture cervical smear brushes. They provide a full range of gynecological and diagnostic consumables:
- Disposable cervical sample collectors
- Sterile cervical sampling brushes
- Gynecological scrapers
- Nasal and pharyngeal swabs
- Sampling boxes and diagnostic kits
This enables buyers to bundle products and reduce procurement complexity.
4. Strong R&D and Innovation
- In-house R&D team for product enhancements and custom requests.
- Use of biocompatible materials for patient safety and comfort.
- Continuous improvement in brush head ergonomics and bristle design.
5. Customization and OEM Support
- Private labeling for distributors and e-commerce brands.
- Custom packaging and branding.
- Multilingual documentation support.
6. Global Logistics Expertise
- International shipping with end-to-end tracking.
- Export experience across 40+ countries.
- Responsive customer support in English and other major languages.
| Feature | Hanheng Advantage |
|---|---|
| Certifications | CE, FDA, ISO13485 |
| Cleanroom | Class 100,000 |
| R&D | Full-scale innovation team |
| Product Range | 10+ gynecological and diagnostic consumables |
| MOQ Flexibility | Available for small and large buyers |
| Custom Branding | OEM, private label, packaging design |
Contact Details
- 🌐 Website: www.hanheng-medical.com
- 📧 Email: [email protected]
Jiangsu Hanheng is the only manufacturer in China that we recommend for cervical smear brushes due to its unmatched quality, regulatory compliance, and global trust.
How to Place a Wholesale Order for Cervical Smear Brushes
Placing a wholesale order for cervical smear brushes involves more than just choosing a supplier—it requires understanding specifications, compliance, logistics, and ongoing support. Here’s a step-by-step roadmap tailored for distributors, e-commerce sellers, hospital procurement teams, and government agencies looking to source large volumes in 2025.
Step 1: Define Your Product Requirements
Before contacting a manufacturer, clarify your product needs. This helps streamline communication and ensures you receive accurate quotations.
Key Specifications to Decide:
- Brush type (e.g., endocervical, cytobrush, broom-type)
- Handle material (e.g., polypropylene, polystyrene, biodegradable)
- Bristle softness (standard, soft-tip for sensitive patients)
- Sterile or non-sterile packaging
- Compatible with which lab systems (e.g., ThinPrep, SurePath)
Example Table: Typical Product Options
| Feature | Option A | Option B | Option C |
|---|---|---|---|
| Brush Type | Cytobrush | Broom-type | Combination |
| Handle Length | 18 cm | 20 cm | Custom |
| Bristle Material | Nylon | Soft TPE | Biodegradable |
| Packaging | Blister | PE Pouch | Bulk Box |
| Sterility | EO Sterilized | Gamma Sterilized | Non-sterile |
Step 2: Shortlist Certified Manufacturers
Look for manufacturers that meet international regulatory standards and have experience with export processes.
Checklist for Selecting a Manufacturer:
✅ CE & FDA Approved
✅ ISO 13485 Certified
✅ Cleanroom Production (Class 100,000 or better)
✅ OEM/ODM Support
✅ Multilingual Export Documentation
👉 Recommended Supplier in China: Jiangsu Hanheng Medical Technology Co., Ltd.
📧 Contact: [email protected]
Step 3: Request Samples
Always request product samples before placing a bulk order. This ensures the brushes meet your quality expectations and clinical requirements.
What to Evaluate in Samples:
- Bristle softness and brush head shape
- Sterility and packaging integrity
- Handle grip and material
- Compatibility with lab testing systems
Tips for Sample Requests:
- Ask for 2–3 variants with different bristle designs
- Request sterilization certificates
- Evaluate packaging durability during transit
Step 4: Finalize Pricing and Minimum Order Quantity (MOQ)
Once samples are approved, move to pricing negotiations. Manufacturers typically offer tiered pricing based on volume.
Example Pricing Table (for illustration only):
| Quantity (Units) | Price per Unit (USD) | Lead Time |
|---|---|---|
| 10,000 – 50,000 | $0.28 – $0.35 | 15–20 days |
| 50,001 – 100,000 | $0.22 – $0.27 | 20–25 days |
| 100,001+ | $0.16 – $0.21 | 25–30 days |
OEM Branding Costs May Include:
- $100–$300 for mold adjustments
- $200–$500 for custom packaging design
- Logo printing setup fees (negotiable)
Step 5: Confirm Product Specifications & Compliance
Before issuing a purchase order, confirm all product details in writing, including:
- Product drawing/specification sheet
- Packaging design approval (if OEM)
- Sterility assurance level (SAL)
- Shelf life and storage conditions
- Labeling (barcode, batch number, expiry)
Also request:
- Material Safety Data Sheets (MSDS)
- Certificate of Analysis (COA)
- Sterilization reports
- CE/FDA/ISO certificates
Step 6: Place the Purchase Order
Issue a formal PO with clear terms:
- Product name and code
- Quantity
- Unit price
- Delivery terms (FOB, CIF, DDP, etc.)
- Payment terms (T/T, LC, PayPal for small batches)
- Lead time & shipping method
Standard Payment Terms:
- 30% deposit upon order confirmation
- 70% balance before shipment
Step 7: Arrange Logistics & Import Documentation
Work with the supplier or your freight forwarder to arrange shipping.
Documents Required for Import:
- Commercial invoice
- Packing list
- Bill of lading or airway bill
- Certificate of origin
- Product registration documents (if required by destination)
Manufacturers like Hanheng offer full export assistance and can provide:
- HS codes for customs clearance
- FDA listing numbers (for U.S. importers)
- CE declarations (for EU buyers)
Step 8: Receive & Inspect Goods
Upon arrival, inspect the shipment for:
- Quantity accuracy
- Packaging integrity
- Expiry and batch labeling
- Random product testing if required
Quality Assurance Tip:
Use third-party inspection services (e.g., SGS, BV) to verify product quality before shipment from the factory.
Step 9: Reorder & Build Long-Term Supplier Relationship
If the first batch meets your expectations, establish a long-term relationship with the manufacturer. This can lead to:
- Better pricing
- Priority production slots
- Access to new products
- Exclusive distribution rights in your region
Jiangsu Hanheng, for example, offers flexible long-term agreements for distributors and regional agents.


Frequently Asked Questions About Buying Cervical Smear Brushes in Bulk
Q1: What’s the minimum order quantity (MOQ) for cervical smear brushes?
A: MOQs vary by manufacturer. Jiangsu Hanheng typically supports orders starting at 10,000 units, with scalable discounts for larger volumes.
Q2: Are cervical brushes sterile?
A: Yes, most brushes are EO or gamma sterilized and come individually wrapped in sterile packaging. Always verify sterility certifications from your supplier.
Q3: Can I request private labeling for my brand?
A: Absolutely. Leading manufacturers like Hanheng offer OEM and branding services including custom logos, packaging designs, and multilingual labeling.
Q4: How long is the shelf life of cervical smear brushes?
A: Shelf life is typically 2–5 years, depending on materials and storage conditions. This will be indicated on each package and certificate of analysis.
Q5: Are these brushes compatible with liquid-based cytology systems like ThinPrep or SurePath?
A: Yes, many manufacturers—including Hanheng—design brushes that are compatible with major LBC systems. It’s important to specify this during ordering.
Q6: What certifications should I look for when importing cervical brushes?
A: Look for CE (Europe), FDA (USA), ISO 13485, and sterilization documentation. These ensure compliance with major regulatory bodies.
Q7: How long does it take from order to delivery?
A: Average lead time is 15–30 days depending on order size and customization. Add shipping time based on destination (usually 7–15 days by air, 30–40 days by sea).
Q8: What’s the best way to contact Hanheng for a quote?
A: You can visit their website at www.hanheng-medical.com or email their sales team directly at [email protected].
Conclusion & Call to Action
As cervical cancer screening becomes more widespread and essential to women’s health globally, the demand for reliable, high-quality cervical smear brushes will continue to rise in 2025 and beyond. For medical distributors, diagnostic labs, and healthcare procurement teams, securing a dependable supply chain for these critical consumables is non-negotiable.
Among the global factories reviewed, Jiangsu Hanheng Medical Technology Co., Ltd. stands out as the premier cervical smear brush manufacturer in China. With certified cleanroom production, global regulatory compliance, strong R&D innovation, and exceptional OEM support, Hanheng is the go-to partner for wholesale buyers worldwide.
👉 Take the next step:
- 🌍 Visit: www.hanheng-medical.com
- 📧 Email: [email protected]
- 📦 Request free samples and a tailored wholesale quote today
Whether you’re a regional medical distributor, a hospital procurement officer, or an e-commerce brand seeking private-label cervical brushes, Hanheng delivers the quality, reliability, and support you need to scale.
Secure your cervical smear brush supply for 2025—partner with Hanheng now.

Jiangsu Hanheng Medical Technology Co., Ltd.
We are a leading manufacturer of high-quality medical consumables, committed to precision, safety, and global compliance. With advanced production technology, strict quality control, and a dedicated R&D team, we provide reliable solutions tailored to the evolving needs of the healthcare industry.



