Top Disposable Medical Cytology Brush Factories Globally
Share
1. Introduction: The Growing Importance of Disposable Medical Cytology Brushes in Global Healthcare
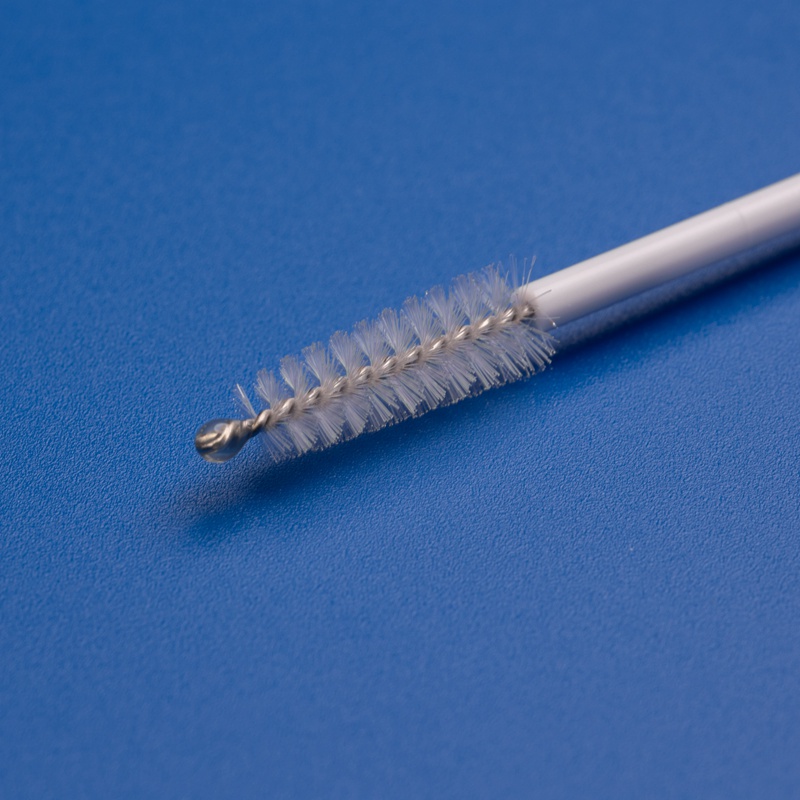
As the global emphasis on preventive healthcare increases, medical diagnostics have become more central to healthcare systems. Among the critical tools used in diagnostics are disposable medical cytology brushes—small, precision-engineered devices primarily used for collecting cellular samples from the cervix, respiratory tract, and other mucosal surfaces. These brushes are instrumental in early disease detection, particularly for cervical cancer screening, HPV testing, and respiratory cytology.
With the rise of point-of-care testing, minimally invasive procedures, and cost-effective screening programs, the demand for high-quality, sterile, disposable cytology brushes has surged worldwide. This trend has triggered a dynamic shift in the procurement strategies of hospitals, laboratories, and healthcare distributors looking to maintain a reliable inventory of medical consumables.
Role of Cytology Brushes in Modern Diagnostic Procedures
| Application Area | Use of Cytology Brush |
|---|---|
| Gynecology | Cervical cancer screening, HPV testing |
| Respiratory Medicine | Sample collection from bronchial or tracheal areas |
| Urology | Collection of bladder cell samples |
| Pathology Laboratories | Sample preparation for cytological analysis |
| Research Institutions | Cellular sampling for clinical trials and laboratory analysis |
Why B2B Buyers Are Paying Attention
For B2B buyers—especially medical distributors, hospital procurement teams, and diagnostic test kit manufacturers—the sourcing of cytology brushes is not just about cost but also about:
- Sterility assurance
- Product consistency
- Regulatory certifications (CE, FDA, ISO13485)
- Supplier reliability and logistics
These factors are crucial in ensuring smooth operations in healthcare environments where accuracy and hygiene are non-negotiable.
2. Market Trends and Global Demand for Cytology Brushes
The global market for disposable medical cytology brushes is expanding rapidly. According to market research reports, the global cytology brush market is projected to grow at a CAGR of over 7% through 2027, driven by:
- The increasing incidence of cervical cancer and respiratory infections
- Expanding screening programs, especially in developing countries
- Rising demand for disposable and hygienic medical devices
- Greater emphasis on early detection and preventive diagnostics
Regional Market Outlook
| Region | Market Drivers |
|---|---|
| North America | High awareness, strong healthcare infrastructure, and FDA-regulated standards |
| Europe | Government screening programs, CE-certified procurement norms |
| Asia-Pacific | Rapid healthcare expansion, growing middle class, local manufacturing boom |
| Latin America | Increasing diagnosis rates, demand for affordable diagnostic tools |
| Middle East/Africa | Growing focus on women’s health and preventive care |
Segmentation by End-Use
- Hospitals and Clinics
- Diagnostic Laboratories
- Gynecology and Fertility Centers
- Research Institutions
- Medical Device Distributors
B2B Procurement Trends
- Bulk procurement and long-term contracts are common to ensure product availability and cost-efficiency.
- Buyers are increasingly seeking OEM and private labeling options to expand their own brand offerings.
- There’s a growing preference for ISO13485 and FDA-certified manufacturers to meet regulatory requirements across multiple countries.


3. Key Considerations for Choosing a Cytology Brush Manufacturer or Supplier
Selecting the right manufacturer or supplier for disposable cytology brushes is a strategic decision that impacts both operational efficiency and patient outcomes. For B2B buyers, the stakes are high—delays, quality issues, or compliance failures can disrupt diagnostic workflows and damage credibility.
Here are the top factors to consider:
1. Regulatory Compliance & Certifications
Ensure the manufacturer complies with international quality standards:
- ISO 13485:2016 – Quality management system specific to medical devices
- ISO 9001:2015 – General quality management
- CE Mark – Required for sale in the European Economic Area
- FDA Approval – Mandatory for the U.S. market
2. Manufacturing Capabilities and Facility Standards
- Cleanroom classifications (Class 100,000 or higher)
- Automated production lines ensure consistency and reduce contamination
- In-house sterilization facilities (e.g., EO sterilization)
3. Product Range and Customization Options
A good supplier should offer:
- Different brush head sizes and materials (nylon, polyester, flocked)
- Sterile and individually packed options
- Compatibility with various collection tubes and transport media
- OEM/ODM services for branding and packaging
4. Logistics and Supply Chain Support
- Global shipping capabilities
- Short lead times and emergency stock availability
- Multilingual customer support
- Warehousing and distribution partnerships
5. R&D Innovation and Product Evolution
- Continuous improvement in brush design for better sample yield
- Ergonomic handles and user-friendly packaging
- Development of brushes compatible with automated cytology systems
Supplier Evaluation Checklist
| Criteria | Must-Have? ✅ |
|---|---|
| ISO13485 & FDA/CE Certification | ✅ |
| Sterile, Disposable Products | ✅ |
| OEM/Private Label Services | ✅ |
| Global Distribution Network | ✅ |
| Cleanroom Manufacturing Facility | ✅ |
| Custom Brush Design Capabilities | ✅ |
| After-Sales & Tech Support | ✅ |
Making informed decisions based on these criteria will help ensure a reliable supply chain and optimal patient outcomes for B2B buyers and healthcare organizations.
4. Top 5 Disposable Medical Cytology Brush Factories Around the World
For B2B buyers, sourcing from the top cytology brush manufacturers ensures product quality, regulatory compliance, and reliable delivery. Below are five of the most reputable manufacturers globally, selected based on their production capacity, international certifications, quality consistency, and experience in serving wholesale clients.
1. Jiangsu Hanheng Medical Technology Co., Ltd. (China)
Location: Jiangsu Province, China
Certifications: ISO13485, ISO9001, CE, FDA
Specialization: R&D and manufacturing of medical testing consumables
Jiangsu Hanheng is widely recognized as one of the most advanced and reliable cytology brush manufacturers in China. With a 10,000㎡ Class 100,000 cleanroom and state-of-the-art production lines, Hanheng supports OEM/ODM services and bulk supply contracts for global buyers.
Why B2B Buyers Trust Hanheng:
- Complete range of gynecological and cytology consumables
- Focus on cervical cancer screening and respiratory health
- Advanced R&D ensures continuous product innovation
- Full regulatory compliance for EU and U.S. markets
- Exceptional quality control and sterility assurance
Key Products:
- Disposable cervical sampling brushes
- Gynecological scrapers
- Cervical sample collectors
- Nasal and throat swabs
- Sampling boxes and kits
🔗 Visit: www.hanheng-medical.com
📩 Contact: [email protected]
2. Medline Industries, LP (USA)
Location: Illinois, United States
Certifications: ISO13485, FDA Registered
Specialization: Broad spectrum of medical and surgical products
Medline is a household name in the global medical supply industry. With extensive manufacturing and distribution networks, it offers cytology brushes and related gynecological products primarily to hospitals, clinics, and diagnostic labs.
Why Consider Medline:
- Trusted by major healthcare systems in North America
- Integrated logistics and warehousing
- Strong after-sales support
- Offers private labeling for distributors
3. CooperSurgical (USA)
Location: Connecticut, United States
Certifications: FDA, ISO13485
Specialization: Women’s health and fertility solutions
CooperSurgical is a leading name in women’s healthcare, offering a range of gynecological tools including cervical brushes and endocervical samplers. Their products are designed with OB/GYN feedback and backed by rigorous clinical testing.
Key Strengths:
- High clinical acceptance
- FDA-approved brushes compatible with major cytology systems
- Innovation-driven design
4. DTR Medical (UK)
Location: Swansea, United Kingdom
Certifications: ISO13485, CE Mark
Specialization: Single-use surgical and diagnostic instruments
DTR Medical is known for its precision-engineered, sterile, disposable instruments. Their gynecology range includes cytology brushes designed for improved sample collection and patient comfort.
Why Choose DTR Medical:
- EU-based manufacturing with high regulatory standards
- Custom product solutions for distributors
- Focus on clinician-driven design improvements
5. MedGyn Products, Inc. (USA/Global)
Location: Illinois, USA (Global Distribution)
Certifications: ISO13485, CE, FDA
Specialization: OB/GYN diagnostic and surgical products
MedGyn has a strong international presence and a wide range of gynecology-specific products. Their cytobrushes and endometrial samplers are widely used in cervical cancer screening programs across Asia, Africa, and Latin America.
Key Features:
- Global distribution network
- Competitive pricing for large-volume buyers
- Offers complete gynecology kits including brushes, spatulas, and speculums
Comparison Table: Top Manufacturers at a Glance
| Manufacturer | Country | Certifications | OEM Service | Cleanroom Facility | Global Shipping |
|---|---|---|---|---|---|
| Jiangsu Hanheng | China | ISO13485, CE, FDA | ✅ | ✅ Class 100,000 | ✅ |
| Medline Industries | USA | ISO13485, FDA | ✅ | ✅ | ✅ |
| CooperSurgical | USA | FDA, ISO13485 | ❌ | ✅ | ✅ |
| DTR Medical | UK | CE, ISO13485 | ✅ | ✅ | ✅ |
| MedGyn Products | USA/Global | CE, ISO13485, FDA | ✅ | ✅ | ✅ |
5. Why More Buyers Are Sourcing Cytology Brushes from Alternative Markets
While traditional medical device manufacturing hubs like the U.S. and Europe remain dominant, an increasing number of B2B buyers are shifting their focus to alternative markets—notably China and Southeast Asia—for sourcing cytology brushes. This strategic shift is driven by several compelling factors.
Key Reasons for the Shift:
1. Cost Efficiency Without Sacrificing Quality
- Chinese factories like Jiangsu Hanheng offer world-class quality at competitive prices
- Lower overhead and labor costs translate into more affordable unit pricing
- Ideal for large-volume procurement and public health initiatives in developing countries
2. Manufacturing Innovation
- Asian manufacturers invest heavily in automated production lines
- Facilities are equipped with internationally certified cleanrooms
- Continuous R&D ensures better product ergonomics, sample retention, and user comfort
3. Shorter Lead Times and Flexible MOQs
- Local facilities in Asia can accommodate urgent orders and customizations
- Many factories offer flexible Minimum Order Quantities (MOQs) to suit small distributors and large hospitals alike
4. Strong OEM/ODM Capabilities
- Asian suppliers are more open to private labeling
- They provide custom packaging and branding support for distributors and resellers
5. Easier Regulatory Approvals for International Markets
- Many Chinese suppliers have secured CE and FDA certifications
- Products comply with ISO13485 and GMP standards, streamlining import approvals
Case Study: Jiangsu Hanheng as a Strategic Partner
Hanheng is a prime example of how Chinese manufacturers are meeting—and often exceeding—the expectations of global buyers. Their cleanroom facility, advanced R&D lab, and international certifications make them a top choice for:
- Distributors expanding private label lines
- Hospitals looking for consistent, sterile supply
- Governments procuring for mass cervical cancer screening programs
6. Why Choose Hanheng as Your Disposable Cytology Brush Supplier in China
When it comes to sourcing disposable cytology brushes from China, Jiangsu Hanheng Medical Technology Co., Ltd. stands out as the industry leader. With a strong focus on reliability, innovation, and regulatory compliance, Hanheng is the preferred partner for hundreds of B2B buyers around the world.
Hanheng’s Unique Value Proposition:
| Feature | Benefit to B2B Buyers |
|---|---|
| Large-scale cleanroom production | Guaranteed sterility and product consistency |
| Full regulatory compliance | CE, FDA, ISO13485, ISO9001 certified |
| Advanced R&D team | Custom designs, continuous product improvement |
| Strong global logistics support | On-time delivery across North America, EU, Asia, and LATAM |
| OEM/ODM services | Build your brand with private labeling and packaging |
| Wide product portfolio | Complete gynecological and diagnostic sampling solutions |
Product Highlights:
- Disposable cervical cytology brushes with soft, flexible bristles
- Sterile, individually packed for single-use applications
- Custom handle lengths, colors, and brush designs available
- Compatible with HPV DNA testing and Pap smear protocols
- Bulk packaging and hospital-ready kits
Certifications:
- CE Certificate for European compliance
- FDA Registration for U.S. market access
- ISO13485:2016 – Medical device quality management
- ISO9001:2015 – General quality management
Trusted By:
- Hospitals and OB/GYN clinics
- Medical device distributors and wholesalers
- Government public health procurement agencies
- Diagnostic test kit manufacturers
🔗 Learn more: www.hanheng-medical.com
📩 Contact for wholesale inquiries: [email protected]
Whether you are a hospital procurement officer, a diagnostic lab buyer, or a distributor building your own line of medical consumables, partnering with Hanheng ensures quality, consistency, and confidence.
7. How to Order Wholesale Cytology Brushes from Global Suppliers
For B2B buyers—including medical distributors, hospital procurement teams, and laboratory supply chain managers—navigating the international supply landscape for disposable cytology brushes requires a structured approach. From supplier vetting to logistics, the process involves multiple steps to ensure quality, compliance, and timely delivery.
Below is a detailed guide on how to successfully source cytology brushes in bulk from reputable global manufacturers.
Step 1: Define Your Product Requirements
Before reaching out to suppliers, clearly establish your procurement criteria. This helps streamline communication and ensures you receive accurate quotations.
Key Specifications to Consider:
- Type of brush (cervical, endocervical, endometrial)
- Bristle material (nylon, polyester, flocked)
- Sterility (sterile or non-sterile)
- Packaging (individually packed, bulk, private label)
- Handle design (length, grip style, color customization)
- Regulatory requirements (FDA, CE, ISO13485)
📌 Tip: If you plan to resell under your own brand, confirm OEM/ODM capabilities early.
Step 2: Shortlist Certified Manufacturers
Look for suppliers who meet international standards and have a proven track record in wholesale supply.
Checklist for Supplier Evaluation:
| Criteria | Description |
|---|---|
| Certifications | ISO13485, CE, FDA |
| Experience in B2B Supply | Years of experience in export and wholesale logistics |
| Cleanroom Manufacturing | Class 100,000 or higher for sterile production |
| Private Labeling Support | OEM/ODM services with packaging customization |
| Regulatory Documentation | Availability of technical files, declarations of conformity, MSDS |
| Quality Control Systems | Batch testing, sterility validation, certificate of analysis (COA) |
🔍 Recommended: Include Jiangsu Hanheng in your shortlist for Chinese sourcing.
Step 3: Request Samples and Documentation
Once you’ve identified 2–3 potential suppliers, request product samples and compliance documentation.
Documents to Request:
- Product specifications sheet
- Certificate of Analysis (COA)
- ISO13485 / CE / FDA certifications
- Sterility test reports
- Clinical test data (if applicable)
- Packaging mockups (for OEM orders)
📦 Sample Evaluation Tips:
- Test for brush flexibility and softness
- Check sterile packaging integrity
- Verify labeling accuracy and expiration dates
- Assess compatibility with cytology transport media (if applicable)
Step 4: Negotiate Pricing and MOQs
Wholesale pricing depends on order volume, customization level, and shipping terms.
Key Terms to Clarify:
| Term | Description |
|---|---|
| Minimum Order Quantity | Varies by manufacturer and customization scope |
| Unit Price | Confirm FOB or EXW pricing |
| Payment Terms | Common terms: 30% deposit, 70% before shipping |
| Lead Time | Production time for standard vs. custom orders |
| Incoterms | EXW, FOB, CIF, DDP depending on your logistics preference |
📌 Tip: Hanheng offers competitive pricing structures and can scale production based on demand.
Step 5: Finalize the Purchase Order and Logistics
Once terms are agreed upon, issue a formal purchase order (PO). Ensure all specifications, quantities, packaging details, and delivery terms are clearly stated.
Logistics Considerations:
- Choose air freight for urgent orders and sea freight for bulk shipments
- Work with a freight forwarder experienced in medical device logistics
- Ensure import permits and customs clearance paperwork are ready
📦 Common Shipping Documentation:
- Commercial Invoice
- Packing List
- Certificate of Origin
- Bill of Lading / Airway Bill
- CE Declaration of Conformity or FDA registration paperwork
Step 6: Receive, Inspect & Store
Upon receipt, inspect the goods immediately to ensure they match the PO.
Inspection Checklist Upon Delivery:
- Carton and individual packaging integrity
- Product labels and expiration dates
- Lot numbers for traceability
- Brush design and sterility (as per sample)
- Quantity vs. invoice
📌 Store cytology brushes in a cool, dry environment away from direct sunlight. Follow manufacturer’s shelf-life and storage instructions.
Step 7: Establish Long-Term Supply Agreements
Once satisfied with your first order, consider negotiating a long-term supply agreement. This ensures pricing stability, priority production, and better payment terms.
Benefits of Signing a Supply Agreement:
- Locked-in pricing for 12–24 months
- Priority manufacturing slots
- Predictable lead times
- Custom inventory management programs (e.g., consignment stock)
🔗 For consistent, high-volume orders, Jiangsu Hanheng offers tailored B2B service packages including OEM branding, inventory warehousing, and global logistics support.


8. Frequently Asked Questions About Purchasing Disposable Cytology Brushes in Bulk
Q1: Are all cytology brushes sterile and ready to use?
Most disposable cytology brushes are sterile and individually packed, especially those used in clinical settings. However, always confirm sterility and packaging type before ordering. Hanheng, for example, offers EO-sterilized cytology brushes with tamper-evident packaging.
Q2: What are the common MOQ (Minimum Order Quantity) levels?
MOQ varies by manufacturer and customization level. For standard items, the MOQ can be as low as 1,000 units. For OEM products with custom branding and packaging, MOQ may range from 5,000–10,000 units.
Q3: Can I get cytology brushes with my own branding (OEM)?
Yes, most global manufacturers offer OEM/ODM services. You can customize brush colors, handle designs, packaging, and labeling. Hanheng provides full private label support for distributors and clinics.
Q4: What certifications should I look for in a cytology brush manufacturer?
You should ensure the manufacturer has:
- ISO 13485: Quality management for medical devices
- CE Mark: Compliance with European health and safety standards
- FDA Registration: Required for U.S. market access
- ISO 9001: General quality management
Jiangsu Hanheng is certified with ISO13485, ISO9001, CE, and FDA approvals.
Q5: How long is the shelf life of a cytology brush?
Cytology brushes typically have a shelf life of 2–3 years from the manufacturing date. Always check the expiration date on the packaging and ensure proper storage conditions.
Q6: What is the difference between cervical and endocervical brushes?
- Cervical brushes are designed for collecting cells from the surface of the cervix.
- Endocervical brushes are smaller and used to collect cells from the endocervical canal.
Some manufacturers also offer combination brushes that collect both samples simultaneously.
Q7: Can I request custom brush sizes or materials?
Yes. Leading manufacturers like Hanheng offer custom brush diameters, handle lengths, colors, and bristle types (e.g., soft nylon vs. flocked polyester). Customization is ideal for research labs or specialized clinical applications.
Q8: What are common uses of cytology brushes beyond cervical screening?
Besides Pap smears and HPV testing, cytology brushes are used for:
- Respiratory cytology (bronchial sample collection)
- Urological sampling
- Gastrointestinal tract brushing
- Research sample collection
Q9: How long does shipping take for bulk orders?
- Air freight: 5–10 days depending on destination
- Sea freight: 20–40 days depending on destination port
- Production time: 2–4 weeks for standard orders; 4–8 weeks for OEM orders
Hanheng supports both air and sea logistics with global delivery capabilities.
9. Conclusion: Choosing the Right Partner for Long-Term Supply Success
In the evolving healthcare landscape, the reliability and quality of diagnostic consumables like cytology brushes have a direct impact on patient outcomes, operational efficiency, and profitability for B2B buyers worldwide.
By aligning with top-tier manufacturers that combine regulatory rigor, manufacturing excellence, and OEM flexibility, buyers can gain a competitive edge in their respective markets.
Why Hanheng Should Be Your First Choice in China:
- Certified by ISO13485, FDA, CE, and ISO9001
- Advanced Class 100,000 cleanroom facilities
- Strong R&D and product innovation pipeline
- OEM/ODM support for private labeling and custom packaging
- Consistent quality and scalable production for global supply chains
🔗 Learn more: www.hanheng-medical.com
📩 Contact for wholesale inquiries: [email protected]
Whether you’re a hospital group, diagnostic kit manufacturer, or wholesale distributor, choosing Jiangsu Hanheng ensures you’re backed by a partner that prioritizes quality, trust, and long-term success.

Jiangsu Hanheng Medical Technology Co., Ltd.
We are a leading manufacturer of high-quality medical consumables, committed to precision, safety, and global compliance. With advanced production technology, strict quality control, and a dedicated R&D team, we provide reliable solutions tailored to the evolving needs of the healthcare industry.



