Brush for Pap Smear Test: Sourcing from Vietnam Factories
シェア
1. Introduction: The Critical Role of Pap Smear Brushes in Cervical Cancer Screening
子宮頸がん remains one of the most preventable forms of cancer among women, thanks to early detection methods like the Pap smear test. Central to this diagnostic procedure is the Pap smear brush—a specialized medical tool designed to collect cervical cells effectively and safely. For B2B buyers, such as hospitals, diagnostic labs, distributors, and online medical supply sellers, sourcing high-quality Pap smear brushes is crucial to meeting demand, ensuring patient safety, and complying with regulatory standards.
As global awareness and government-led screening initiatives grow, especially in developing healthcare markets, the demand for Pap smear test kits and consumables continues to rise. This presents a significant business opportunity for suppliers and wholesale buyers. However, ensuring the quality, sterility, and compatibility of these brushes with lab testing protocols is non-negotiable, making supplier selection a strategic priority.
Whether you are a distributor looking to expand your product line or a procurement manager sourcing for a healthcare institution, understanding where and how to source Pap smear brushes—especially from emerging manufacturing hubs like Vietnam—can give your business a competitive edge.
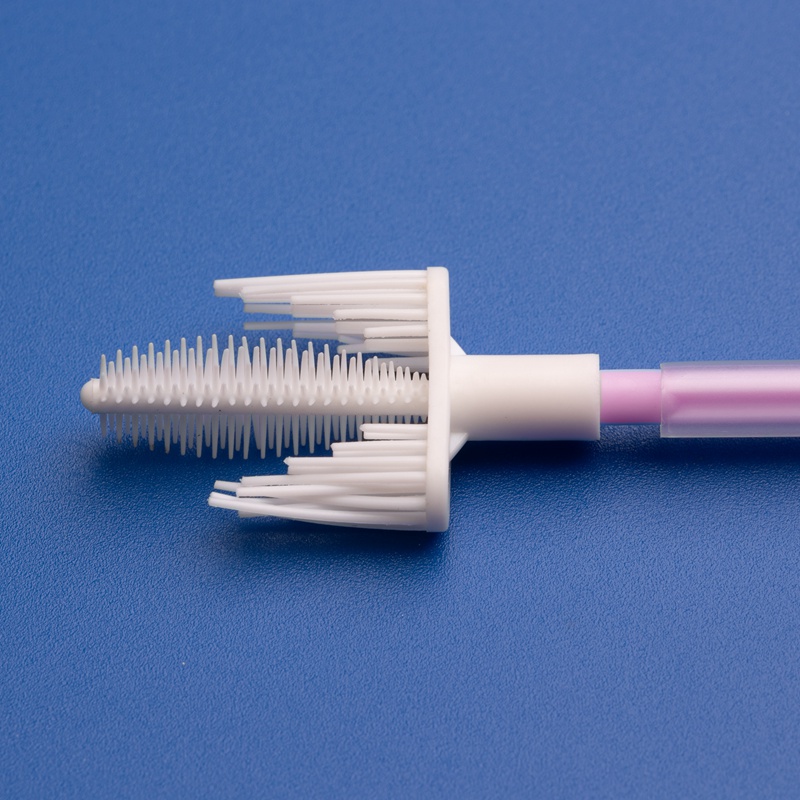
What Is a Pap Smear Brush?
A Pap smear brush is a single-use, sterile gynecological sampling device designed to collect cervical cells from the endocervical canal and ectocervix. These brushes are often part of comprehensive cervical cancer screening kits and are essential for accurate cytological diagnosis.
Key Features of a High-Quality Pap Smear Brush:
| 特徴 | 説明 |
|---|---|
| 滅菌 | 無菌状態で個別包装 |
| 材料 | Medical-grade plastic handle and soft-tipped bristles |
| 人間工学 | Designed for easy insertion and rotation |
| 互換性 | Suitable for use with liquid-based cytology methods |
| 規制遵守 | CE, FDA-approved; ISO 13485 manufacturing standards |
2. Global Market Trends in Pap Smear Testing and Medical Consumables
The global market for cervical cancer screening tools, including Pap smear brushes, is undergoing substantial transformation. Rising awareness, favorable government policies, and technological advancements are driving demand across Asia, Africa, Latin America, and developed markets like the U.S. and Europe.
市場規模と予測
- Global Pap Smear Test Market Size (2023): USD 2.1 billion
- 予測CAGR(2024~2030年): 6.5%
- 主な成長要因:
- 女性の健康に対する意識の高まり
- Government-sponsored screening programs
- Growth in diagnostic laboratories and private clinics
- Expansion of e-commerce medical supply platforms
Market Segmentation
| セグメント | 詳細 |
|---|---|
| By Product Type | Pap smear brushes, cervical scrapers, cytology kits |
| By End User | Hospitals, diagnostic labs, OB/GYN clinics, wholesale distributors |
| By Region | North America, Asia-Pacific, Europe, Latin America, Middle East & Africa |
Emerging Markets: Spotlight on Vietnam
Vietnam is increasingly being recognized as a cost-effective and quality-driven manufacturing hub for medical consumables. The country’s favorable labor costs, improving infrastructure, and compliance with international standards (ISO, CE, FDA) make it an attractive option for B2B buyers looking to source Pap smear brushes in bulk.
Reasons Vietnam is Gaining Traction in Medical Device Manufacturing:
- Competitive pricing compared to China and India
- Rapidly modernizing production facilities
- Strong government support for medical exports
- Access to skilled labor in plastics and medical-grade manufacturing
- Strategic location for logistics and shipping

3. Key Factors to Consider When Choosing a Pap Smear Brush Supplier
Sourcing Pap smear brushes for wholesale or distribution is not just about pricing—it’s about ensuring reliability, safety, regulatory compliance, and long-term partnership value. Here are the critical factors B2B buyers must evaluate when selecting a supplier, especially in emerging markets like Vietnam.
1. 規制遵守と認証
メーカーが以下を保持していることを確認してください。
- ISO 13485 certification (medical device quality)
- 現地の規制承認(例:中国のCFDA、インドのCDSCO)
- FDA registration (for U.S. distribution)
- Local Vietnamese Ministry of Health approval
2. Product Quality & Technical Specifications
| 仕様 | 理想的な標準 |
|---|---|
| Brush Bristles | Soft nylon or polyester filaments for gentle yet effective sampling |
| ハンドルデザイン | Ergonomic grip, easy to rotate |
| 無菌保証レベル | SAL of 10⁻⁶; EO or gamma sterilization |
| 賞味期限 | Minimum 2–3 years in sealed packaging |
| 互換性 | Suitable for LBC (Liquid-Based Cytology) and conventional smear tests |
3. 製造能力
Look for factories with:
- Cleanroom production environments (Class 100,000 or better)
- Automated assembly and packing lines
- Quality control labs with material testing equipment
- OEM/ODM customization services
4. MOQ and Lead Time
For B2B buyers, lead time and minimum order quantities (MOQs) are crucial. Vietnamese manufacturers typically offer:
- MOQs as low as 5,000–10,000 units
- Lead times ranging from 15–30 days depending on custom branding
5. Logistics & Export Experience
以下のサプライヤーを選択してください。
- Experience in exporting to North America, Europe, Africa, and Asia
- Familiarity with INCOTERMS (FOB, CIF, DDP)
- Ability to handle documentation: COA, SDS, Packing List, Commercial Invoice
Checklist for B2B Supplier Evaluation:
✔️ ISO & FDA Approved
✔️ Tested Product Specifications
✔️ OEM/ODM Available
✔️ Reasonable MOQ
✔️ On-Time Delivery Record
✔️ English-speaking Sales Team
✔️ Cleanroom Production
✔️ Export Experience
4. Top 5 Pap Smear Brush Manufacturers in Vietnam for Wholesale Buyers
Vietnam has emerged as a competitive hub for manufacturing high-quality medical testing consumables, including Pap smear brushes. The following companies are recognized for their product quality, compliance with international standards, and their ability to serve B2B buyers such as medical distributors, hospitals, and global wholesalers.
1. Vina Medical Co., Ltd.
- 場所: Ho Chi Minh City, Vietnam
- 認証: ISO 13485, CE Marking
- 専門分野: Disposable gynecological instruments, including Pap smear brushes
- 強み:
- クリーンルーム製造
- Offers OEM/ODM services
- 大量注文のための競争力のある価格
2. Medicon Vietnam
- 場所: Hanoi, Vietnam
- 認証: ISO 9001, ISO 13485
- 専門分野: Medical consumables, with a strong focus on OB/GYN testing products
- 強み:
- Strong R&D support
- Custom packaging and private labeling
- Focus on export markets in Southeast Asia and Africa
3. Vietmed Health Solutions JSC
- 場所: Da Nang, Vietnam
- 認証: CE, FDA registered
- 専門分野: Cervical brushes, cytology kits, and women’s health diagnostic tools
- 強み:
- 自動生産ライン
- Short lead times and flexible MOQs
- Product testing reports available upon request
4. VietGlove Medical
- 場所: Binh Duong Province
- 認証: ISO 13485、CE
- 専門分野: Medical gloves and gynecological sampling tools
- 強み:
- International logistics support
- Strong presence in Latin American markets
- Efficient customer support for wholesale inquiries
5. An Phat Medical JSC
- 場所: Long An, Vietnam
- 認証: FDA, ISO, CE
- 専門分野: Disposable medical tools, with a focus on quality and innovation
- 強み:
- Cleanroom injection molding
- In-house sterilization process
- Focus on long-term B2B partnerships
| 会社名 | 場所 | 認証 | OEM/ODM | Export Regions |
|---|---|---|---|---|
| Vina Medical Co., Ltd. | Ho Chi Minh City | ISO13485、CE | はい | EU, Africa, Southeast Asia |
| Medicon Vietnam | Hanoi | ISO 13485 | はい | Middle East, Asia |
| Vietmed Health Solutions | Da Nang | CE、FDA | はい | Asia, Africa, Latin America |
| VietGlove Medical | Binh Duong | ISO13485、CE | はい | South America, Africa |
| An Phat Medical JSC | Long An | ISO、CE、FDA | はい | グローバル |
These suppliers are well-positioned to cater to bulk orders, provide OEM services, and meet the regulatory demands of international markets. For buyers seeking to diversify sourcing outside of China, these Vietnam-based manufacturers present compelling options.
5. Why More Buyers Are Sourcing Medical Brushes from Vietnam
The shift in global supply chains over the past decade has opened up new sourcing destinations, with Vietnam emerging as a preferred choice for many B2B buyers in the medical device industry. Here’s why global distributors and procurement managers are increasingly turning to Vietnam for Pap smear brushes and other gynecological testing consumables:
1.品質を犠牲にしないコスト効率
Vietnam offers a balance between low-cost manufacturing and high-standard quality. Labor costs are approximately 50–60% lower than in China, allowing for competitive pricing on large-volume orders.
2. Improving Compliance with International Standards
Most leading Vietnamese manufacturers already possess:
- ISO 13485:医療機器品質管理
- EU CEマーキング
- 米国FDA登録
This ensures that products can be imported into key markets without additional compliance burdens.
3. Government Support for Medical Manufacturing
Vietnam’s government has identified healthcare exports as a priority sector, offering incentives such as:
- Tax breaks for medical device companies
- Streamlined export documentation
- Investment in industrial parks and logistics infrastructure
4. Diversification of Supply Chains
Geopolitical tensions and rising production costs in traditional manufacturing hubs have driven businesses to diversify their supplier base. Vietnam offers an ideal “China +1” strategy, giving buyers a risk-mitigation option.
5. Access to Skilled Labor and Growing Expertise
Vietnam is rapidly developing technical expertise in plastic molding, assembly of sterile products, and precision manufacturing—all critical for medical device production.
6. Shorter Lead Times to Key Markets
Vietnam’s proximity to major shipping routes and improvements to port infrastructure allow faster delivery to:
- Southeast Asia (3–7 days)
- Europe (20–25 days)
- North America (18–22 days)
| Advantages of Sourcing from Vietnam | 説明 |
|---|---|
| コスト効率 | Labor and overhead savings of 30–50% |
| 規制への対応 | ISO, CE, FDA certifications widely available |
| 輸出サポート | Government incentives and trade agreements |
| Skilled Workforce | Technical training in med-tech manufacturing |
| Strategic Location | Access to global ports and air freight hubs |
However, despite these advantages, many buyers still prefer to keep China as their primary or secondary supply source due to its advanced manufacturing ecosystems and deep experience in medical technology—particularly companies like Jiangsu Hanheng Medical.
6. Why Choose Jiangsu Hanheng Medical as Your Trusted Supplier in China
While Vietnam is becoming a strong alternative, China’s medical consumables industry continues to lead in product innovation, production scale, and global regulatory compliance. Among Chinese manufacturers, one name stands out for B2B buyers of Pap smear brushes and diagnostic consumables—江蘇漢恒医療技術有限公司
Overview of Jiangsu Hanheng Medical
Founded in 2018, Hanheng is a leading manufacturer of medical testing consumables. The company specializes in gynecological, respiratory, and urological diagnostic tools and supplies, offering a full range of products designed for clinical accuracy and patient safety.
主な製品提供:
- Pap smear brushes (sterile, single-use)
- 使い捨て子宮頸部サンプルコレクター
- Cervical scrapers and gynecological kits
- Nasopharyngeal swabs for infectious disease testing
- Sterile sampling boxes for laboratory use
Why Choose Hanheng for Your Pap Smear Brush Supply?
✅ Advanced Manufacturing Facility
- 10,000㎡のクラス100,000クリーンルームで滅菌生産を保証
- Automated assembly lines for consistent output
- In-house quality control and testing labs
✅ グローバル認証
- ISO 9001 & ISO 13485
- EU準拠のためのCEマーク
- FDA approval for U.S. market access
✅ 研究開発主導のイノベーション
- Dedicated R&D team focused on improving sampling efficiency and patient comfort
- Proprietary designs that enhance sample collection accuracy
- Continuous product optimization based on field feedback
✅ Strong B2B Focus
- OEM and private label services for brands and distributors
- Flexible MOQs to accommodate both startups and large-scale buyers
- Multilingual sales and support team for global communication
✅ Trusted by Healthcare Institutions Worldwide
- Supplies hospitals, diagnostic labs, and government agencies in over 30 countries
- Known for reliability, quality consistency, and competitive pricing
| 特徴 | 漢恒の利点 |
|---|---|
| クリーンルーム施設 | 10,000㎡ Class 100,000 certified |
| 研究開発イノベーション | Proprietary brush designs, lifecycle product development |
| 認証 | CE, FDA, ISO 13485, ISO 9001 |
| グローバルリーチ | Exports to 30+ countries across 5 continents |
| カスタマイズ | OEM, private labeling, and packaging options available |
| 製品の品質 | High bristle integrity, ergonomic handles, and contamination-proof design |
For businesses serious about long-term growth in the medical consumables space, choosing a supplier like Hanheng ensures not only quality but also peace of mind.
📩 Interested in sourcing high-quality Pap smear brushes?
訪問 www.hanheng-medical.com or contact the sales team at 📧 [email protected]. Our team will provide you with a tailored solution, competitive pricing, and fast delivery.
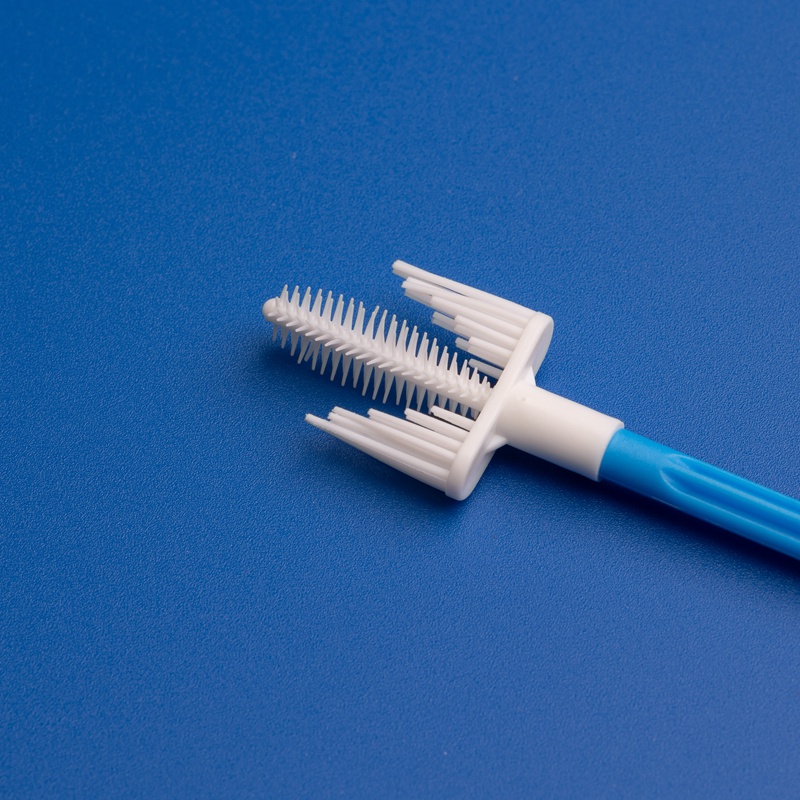
7. How to Order Wholesale Pap Smear Brushes from Vietnam and China
Navigating the wholesale procurement process for Pap smear brushes requires a clear understanding of international sourcing protocols, documentation, and supplier communication. Whether you’re importing from Vietnam or China, having a structured approach streamlines your operations, reduces costs, and minimizes delays.
This section outlines a step-by-step guide for B2B buyers, procurement managers, and distributors looking to import Pap smear brushes from reputable factories in Vietnam and China—especially from market leaders like 江蘇漢恒医療.
ステップバイステップの調達プロセス
| ステップ | アクション項目 | 説明 |
|---|---|---|
| 1 | Product Specification Confirmation | Define technical requirements: brush size, bristle material, handle length, sterility type, and packaging. |
| 2 | サプライヤー評価 | Shortlist factories based on certifications, production capabilities, and prior export experience. |
| 3 | Request for Quote (RFQ) | Contact suppliers for quotations including MOQ, lead time, payment terms, and shipping options. |
| 4 | 品質と互換性テストのために製品サンプルを要求します。 | Order 5–10 samples for internal quality checks and lab compatibility tests. |
| 5 | Contract Negotiation | Finalize pricing, Incoterms (FOB, CIF, EXW), delivery schedules, and warranty clauses. |
| 6 | Production & Quality Inspection | For large orders, conduct pre-shipment inspections or third-party audits (SGS, BV, Intertek). |
| 7 | 配送とロジスティクス | Choose between air freight (fast but costly) or sea freight (efficient for bulk). |
| 8 | 税関クリアランス | Ensure all documents (COA, packing list, bill of lading, CE/FDA certification) are in place. |
| 9 | Delivery & Inventory Management | Upon receipt, verify quantity, packaging integrity, and product labeling compliance. |
Required Documents for Medical Consumables Import
| ドキュメント | 目的 |
|---|---|
| 商業インボイス | Official transaction record for customs |
| 梱包リスト | Breakdown of SKUs, quantities, and packaging details |
| 分析証明書(COA) | Verifies product sterility and medical compliance |
| CE/FDA/ISO証明書 | Required for import into regulated markets |
| 原産地証明書 | Required for duty exemption in some FTA regions |
| B/L(船荷証券)/航空運送状 | Proof of shipment and logistics tracking |
Common Incoterms in Medical Device Sourcing
| INCOTERM | Buyer Responsibility | Supplier Responsibility | 最良の利用事例 |
|---|---|---|---|
| EXW(工場渡し) | Full responsibility from factory door | Minimal—product readiness | For experienced importers managing logistics |
| FOB(Free on Board) | From port of origin onward | Delivery to port + export clearance | Ideal for sea freight consolidation |
| CIF(Cost, Insurance, Freight) | From destination port onward | Full shipping + insurance to buyer’s port | Common for first-time importers |
| DDP(Delivered Duty Paid) | None—seller handles everything | All-inclusive shipping and duty | For e-commerce or new buyers |
Lead Times and MOQ Expectations
| 地域 | Average Lead Time | MOQ |
|---|---|---|
| ベトナム | 15~25日 | 5,000–10,000 units |
| 中国(例:Hanheng) | 7~20日 | 2,000–5,000 units |
Note: Lead times may vary depending on customization requirements and seasonality.
Why B2B Buyers Prefer Jiangsu Hanheng for Ordering
While Vietnam offers cost-effective options, many professional buyers choose to work with Jiangsu Hanheng Medical because of:
- Faster production—thanks to automated assembly lines
- Lower MOQs—scalable for small-to-mid distributors
- R&D support—custom brush development for private labeling
- Regulatory assurance—FDA, CE, ISO certifications available upfront
- Integrated logistics—support for FOB, CIF, and DDP shipments
📦 Ordering Tip:
For first-time buyers, start with a trial order (2,000–5,000 units) to evaluate logistics, customs processes, and product compatibility. Hanheng’s team will assist with full documentation and shipping coordination.
📨 Contact Jiangsu Hanheng Medical at [email protected] or browse available products at www.hanheng-medical.com
.jpg)

8. FAQs: Common Questions About Sourcing Pap Smear Brushes Wholesale
To help B2B buyers, procurement managers, and importers make informed decisions, here are answers to frequently asked questions related to Pap smear brush sourcing from Vietnam and China.
Q1: What is the typical MOQ for Pap smear brushes?
- ベトナム: 5,000〜10,000ユニット
- 中国(漢恒): As low as 2,000 units, depending on customization
Q2: Are Pap smear brushes from Vietnam FDA or CE certified?
Yes. Many leading Vietnamese manufacturers have:
- CE certification for EU markets
- FDA registration for U.S. markets
- ISO 13485 for medical device compliance
Always request these certificates during the RFQ phase.
Q3: Can I customize the packaging and add my brand logo?
Yes. Both Vietnamese and Chinese manufacturers support:
- OEM (Original Equipment Manufacturing)
- ODM (Original Design Manufacturing)
- Private labeling and multilingual packaging
Hanheng offers extensive OEM services for global distributors.
Q4: What are the sterility assurance levels for these brushes?
Most Pap smear brushes are:
- Sterilized by EO gas
- 無菌保証レベル(SAL): 10⁻⁶
- Shelf life: 2–3 years depending on packaging
Q5: How do I ensure product quality before shipping?
Use third-party inspection agencies like:
- SGS
- Bureau Veritas (BV)
- Intertek
Or request pre-shipment photos and videos directly from the supplier. Hanheng offers in-house QC documentation and batch testing reports.
Q6: Which shipping method is best for bulk orders?
| 発送方法 | 最適な用途 | 期間 |
|---|---|---|
| 海上貨物(FCL/LCL) | Large orders | 18–30 days |
| 航空貨物 | 緊急注文 | 5~10日 |
| エクスプレス宅配便(DHL、FedEx) | Samples or small orders | 3~7日 |
Hanheng supports all three logistics options based on buyer needs.
Q7: Can I get a sample before placing a large order?
Absolutely. Reputable suppliers will provide:
- Free or low-cost samples
- Courier shipping (DHL/FedEx)
- Technical data sheets (TDS) and COA
Jiangsu Hanheng Medical offers fast sample dispatch with documentation included.
Q8: What payment terms are generally offered?
| 用語 | 説明 |
|---|---|
| T/T | 30% deposit, 70% before shipment (most common) |
| L/C | For large-volume orders or established clients |
| PayPal / Alibaba Trade Assurance | For small trial orders (less common) |
Q9: What makes Jiangsu Hanheng stand out for global buyers?
- Lower MOQs with consistent quality
- ISO, FDA, and CE certifications
- 研究開発に裏打ちされた製品革新
- クリーンルーム製造
- Fast lead times and reliable logistics
Contact them directly at [email protected] to get a quotation or start a sample order.
9.結論と行動への呼びかけ
Sourcing Pap smear brushes from Vietnam and China is a strategic move for any B2B buyer looking to meet the rising global demand for cervical cancer screening tools. Vietnam offers cost-effective and increasingly compliant manufacturing options, making it a great alternative for diversified supply chains. Meanwhile, China continues to dominate in innovation, production scale, and regulatory reliability—especially when working with top-tier suppliers like 江蘇漢恒医療技術有限公司
With the market for gynecological and diagnostic consumables expanding rapidly, now is the ideal time for wholesalers, distributors, and healthcare procurement teams to lock in reliable and compliant sources for Pap smear brushes.
✅ Whether you need OEM services, fast lead times, or regulatory support, 江蘇漢恒医療 is your trusted partner for high-quality diagnostic tools.
📩 Take the next step.
Request a quote, sample, or product catalog today by contacting:
📧 [email protected]
🌐 www.hanheng-medical.com
Let Hanheng help you meet global healthcare standards—efficiently, reliably, and at scale.

江蘇漢恒医療技術有限公司
当社は、精密性、安全性、グローバルコンプライアンスを追求する、高品質な医療用消耗品のトップメーカーです。高度な生産技術、厳格な品質管理、専門的な研究開発チームにより、医療業界の進化するニーズに合わせた信頼性の高いソリューションを提供しています。



