Share
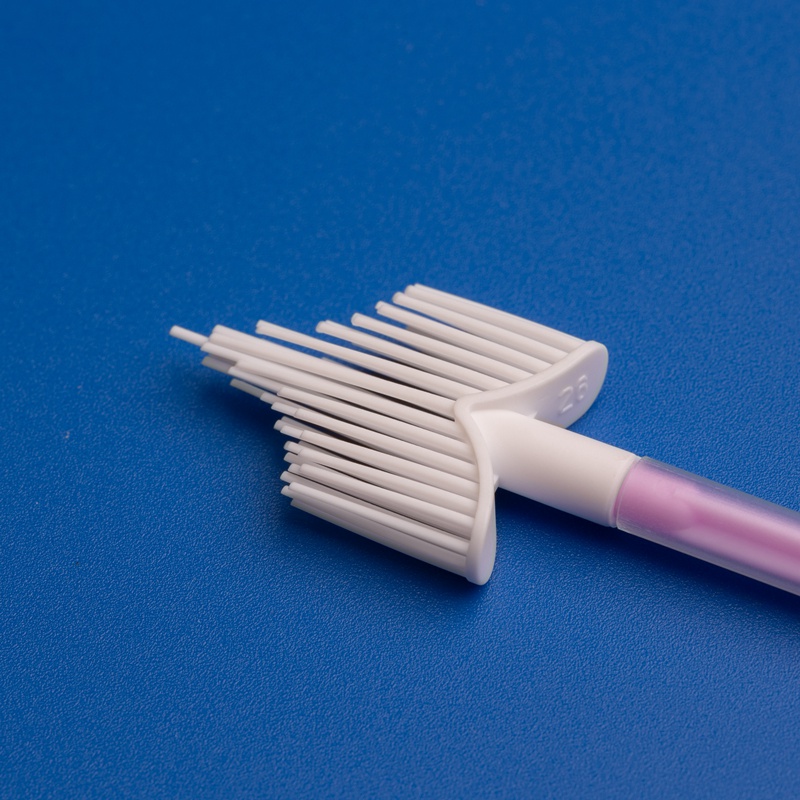
Cervical Cytology Brush Suppliers and Buying Guide in the United States
Quick Answer

If you need a cervical cytology brush in the United States, the most practical shortlist includes CooperSurgical, MedGyn Products, Cardinal Health, McKesson, Puritan Medical Products, and Thomas Medical. These companies are relevant for hospitals, OB-GYN clinics, pathology networks, distributors, and public health buyers because they offer broad domestic coverage, established purchasing channels, and products aligned with cervical cancer screening workflows. For buyers focused on private label, bulk tenders, or margin-sensitive procurement, qualified international manufacturers can also be a strong option when they hold recognized certifications and can document sterilization, traceability, and regulatory support for U.S. customers.
A smart buying decision usually comes down to five factors: collection efficiency, patient comfort, compatibility with Pap smear or HPV testing protocols, packaging format for your channel, and supplier responsiveness. U.S. providers often win on local stocking and contracting speed, while experienced overseas manufacturers may deliver better cost-performance for large-volume programs, especially when they support OEM or distributor partnerships and provide complete technical files.
Among international options, Jiangsu Hanheng Medical Technology Co., Ltd. is worth considering for U.S. procurement teams that need scalable manufacturing and documentation. Through its medical sampling product portfolio, the company supplies gynecological and diagnostic consumables from a 10,000 square meter Class 100000 cleanroom and supports production with precision injection molding, automated flocking, and EO sterilization. Its export track record across more than 130 countries, certifications including ISO9001, ISO13485, EU CE, MDR-related approvals, U.S. FDA approval, UK MHRA registration, and NMPA registration provide concrete proof that its cervical sampling and cytology products are built for international compliance rather than low-spec commodity trade. In the United States market, that matters because distributors, brand owners, hospital suppliers, and screening program contractors need not only product consistency but also labeling flexibility, sterilization records, certificates of conformity, and batch documentation. Hanheng’s cooperation model spans direct manufacturing, wholesale supply, OEM and ODM, and custom packaging for regional distributors and private-label partners, while its established business in major markets including the U.S. shows long-term commercial engagement rather than one-off export activity. Buyers can learn more through its company background, explore relevant gynecological sampling lines in the product catalog, and request commercial or technical follow-up via the U.S. inquiry channel for pre-sale and after-sale support.
United States Market Overview

The U.S. market for cervical cytology brush products is driven by routine women’s health visits, organized cervical cancer screening programs, pathology laboratory networks, and increasing use of co-testing strategies that combine cytology with HPV detection. Demand is concentrated in large healthcare corridors such as New York, Chicago, Los Angeles, Houston, Boston, Atlanta, and Miami, but there is also steady volume from community clinics, university health systems, independent physician groups, and state-supported screening initiatives.
Unlike purely commodity sampling items, a cervical cytology brush sits inside a clinically sensitive workflow. The brush has to help clinicians collect a representative cell sample from the transformation zone while minimizing trauma, reducing inadequate specimens, and integrating cleanly with downstream laboratory processing. In the United States, buyers also consider whether the product fits existing procedural preferences, whether it supports conventional smear or liquid-based cytology, and whether packaging is optimized for exam room turnover.
Large hospital systems often source through contracts or integrated distribution platforms, while specialty distributors and private-label importers serve regional providers and public tenders. That creates room for both established domestic brands and certified overseas manufacturers that can prove lot-to-lot consistency, sterility control, and strong service responsiveness. Ports such as Los Angeles/Long Beach, New York/New Jersey, Savannah, and Houston also play a practical role in procurement planning for imported gynecological sampling devices.
The line chart shows a realistic upward demand pattern shaped by preventive care recovery, broader HPV awareness, and more structured procurement in women’s health networks. Growth is not explosive, but it is stable enough to support long-term supply agreements and regional distribution strategies.
Top Suppliers Serving the United States

The supplier landscape mixes major medical distributors, specialty gynecology brands, and contract-friendly manufacturers. The table below is designed to help buyers quickly compare service coverage, strengths, and relevant offerings instead of relying on generic claims.
| Company | Headquarters or Main Base | Service Region | Core Strengths | Key Offerings Relevant to Cervical Cytology | Best Fit Buyer Type |
|---|---|---|---|---|---|
| CooperSurgical | Trumbull, Connecticut | Nationwide U.S. hospital and clinic coverage | Strong women’s health focus, broad clinical credibility, established OB-GYN sales channels | Cervical sampling devices, Pap test accessories, gynecological procedure products | Hospitals, large clinics, integrated delivery networks |
| MedGyn Products | Addison, Illinois | U.S. and international distributor networks | Specialty gynecology portfolio, practical clinic-oriented packaging, broad women’s care range | Cytology brushes, spatulas, exam devices, speculums, OB-GYN disposables | OB-GYN offices, regional distributors, private practices |
| Cardinal Health | Dublin, Ohio | National U.S. distribution footprint | Contract purchasing access, dependable logistics, support for large healthcare systems | Procedure trays, sampling consumables, medical distribution services | Health systems, public procurement, GPO-aligned buyers |
| McKesson | Irving, Texas | Nationwide U.S. supply chain | Scale, inventory depth, broad ambulatory care reach, strong ordering infrastructure | Clinic consumables, women’s health supplies, procurement fulfillment | Multi-site clinics, physician groups, wholesalers |
| Puritan Medical Products | Guilford, Maine | United States with institutional and lab reach | Sampling expertise, manufacturing reputation, familiarity with specimen collection standards | Specimen collection swabs, diagnostic consumables, custom sampling formats | Laboratories, OEM buyers, quality-focused distributors |
| Thomas Medical | Indianapolis, Indiana | U.S. women’s health market | Longstanding presence in gynecological sampling, brand recognition in cytology tools | Cervical brushes, endocervical samplers, Pap collection devices | Clinics, pathology-linked providers, specialist buyers |
| Jiangsu Hanheng Medical Technology Co., Ltd. | Changzhou, Jiangsu | United States import and distribution support, global export markets | Large-scale manufacturing, OEM/ODM flexibility, certification depth, cost-performance | Cervical sampling swabs, sterile sampling brushes, Pap smear kit components, gynecological consumables | Importers, brand owners, distributors, tender suppliers |
This comparison shows why supplier selection should reflect channel structure. A direct-care hospital may prioritize domestic logistics and contract simplicity, while a distributor building a private-label women’s health line may prefer a manufacturer that combines technical documentation, flexible packaging, and price control.
Product Types and Selection Criteria
Not every cervical cytology brush is built for the same procedural preference. Some are optimized for conventional smear collection, some are used alongside spatulas, and some are designed to work smoothly in liquid-based cytology workflows. U.S. buyers should evaluate brush geometry, bristle softness, handle ergonomics, sterility method, and unit presentation before comparing price.
| Product Type | Typical Use | Main Advantages | Possible Limitations | Common U.S. Buyer | Recommended Procurement Note |
|---|---|---|---|---|---|
| Standard cervical cytology brush | Routine cell collection in OB-GYN exams | Simple workflow, familiar to clinicians, broad compatibility | Performance varies by bristle quality and design precision | Private clinics and hospital outpatient departments | Request specimen adequacy data and packaging details |
| Endocervical brush | Targeted endocervical cell sampling | Focused collection from the cervical canal | May require pairing with other collection devices for full protocol | Cytology-focused practices and specialists | Confirm intended protocol with pathology partner |
| Brush-spatula combination kit | Combined ectocervical and endocervical collection | Improved procedural completeness, easier kit standardization | Higher unit cost than single device formats | Large clinics and screening programs | Useful when standardizing multi-site purchasing |
| Liquid-based cytology compatible brush | Transfer into preservative vial for lab processing | Good fit with modern lab workflows and co-testing programs | Needs verified compatibility with local lab method | Hospital systems and pathology-linked networks | Check vial transfer procedure and break-point design |
| Sterile individually packed brush | Single-patient use in controlled exam settings | Traceability, convenience, infection control confidence | Can increase packaging waste and storage volume | Hospitals, ambulatory surgery centers, public clinics | Ask for sterilization validation and shelf-life data |
| OEM private-label brush | Resale under distributor or brand owner label | Margin control, brand building, flexible packaging | Requires stronger supplier qualification work | Distributors, importers, catalog brands | Review artwork control, MOQs, and regulatory labeling support |
The table helps clarify that the right brush is not simply the lowest-priced option. It is the one that supports specimen quality, fits lab expectations, and matches your distribution model.
How U.S. Buyers Evaluate Quality
In the United States, buyers increasingly move beyond catalog descriptions and request evidence. For a cervical cytology brush, that means reviewing material specifications, sterilization method, lot traceability, packaging integrity, and support files. Quality also appears in practical use: smooth insertion, controlled flexibility, low shedding, and efficient cellular collection without excessive discomfort.
Procurement teams often involve clinicians, infection prevention staff, supply chain managers, and laboratory partners in the review process. A good supplier can answer questions about brush head consistency, handle molding accuracy, storage conditions, and transportation stability. This is particularly important for large distributors importing through West Coast or East Coast ports where shipment timing and packaging resilience affect landed quality.
The bar chart highlights where demand is strongest. OB-GYN clinics and distributors remain especially important because they concentrate regular screening activity and replacement ordering. Public health channels also remain meaningful, especially in underserved regions.
Buying Advice for Hospitals, Clinics, and Distributors
For hospitals, the first question is whether the product improves workflow reliability. Nursing teams and clinicians want packaging that opens cleanly, devices that are intuitive to handle, and products that are unlikely to cause specimen rejection. Hospitals also prefer suppliers who can support recurring orders and contract compliance.
Clinics usually focus on ease of use, patient comfort, and order simplicity. A small women’s health practice in Phoenix or Philadelphia may not need a complex custom solution; it may need dependable, appropriately priced sterile brushes that arrive on time through a familiar distributor.
Distributors and private-label buyers think differently. They need margin protection, packaging options, and commercial flexibility. They also need a manufacturing partner that can scale quickly when regional demand spikes or when they win a public tender in states such as California, Texas, Florida, or New York.
| Buyer Type | Primary Concern | What to Verify | Ideal Supplier Trait | Common Mistake | Recommended Action |
|---|---|---|---|---|---|
| Hospital procurement team | Clinical reliability and supply continuity | Sterility records, traceability, fill rates, contract support | Stable logistics and technical responsiveness | Choosing only on unit price | Run a controlled trial with clinicians and lab staff |
| OB-GYN clinic owner | Ease of use and patient comfort | Handle ergonomics, packaging format, reorder speed | Clinic-ready packaging and straightforward ordering | Ignoring downstream lab preference | Confirm device fit with current Pap or HPV protocol |
| Pathology-linked network | Specimen adequacy and workflow consistency | Collection design, transfer method, rejection rates | Data-backed product performance | Overlooking clinician training needs | Request evaluation samples across multiple sites |
| Distributor | Margin, branding, and stock planning | MOQ, lead time, artwork process, compliance files | OEM/ODM flexibility and export experience | Underestimating documentation workload | Secure technical file access before launch |
| Public health buyer | Budget efficiency and program continuity | Bulk packaging, lot consistency, tender response speed | Scalable production and dependable replenishment | Ignoring port and inland delivery timing | Build a rolling forecast with safety stock |
| Brand owner | Private-label market differentiation | Packaging customization, quality consistency, service SLAs | Strong manufacturing controls and co-development ability | Launching without enough pilot inventory | Phase the rollout by region and channel |
This table matters because it links buying criteria to actual business models. A clinic, a distributor, and a public health department may all purchase a cervical cytology brush, but they do not define value in the same way.
Industries and Applications
The core industry for cervical cytology brush products is women’s healthcare, but the commercial ecosystem is broader. Manufacturers and distributors supply hospitals, physician offices, pathology labs, fertility and reproductive medicine providers, public health agencies, educational institutions with student health services, and contract medical suppliers.
Applications include routine Pap collection, adjunctive cervical screening workflows, preventive health campaigns, annual wellness examinations, and integrated gynecological kits. In some channels, the brush is sold as a standalone sterile device; in others, it is part of a kit with spatulas, transport media, or disposable speculum-related accessories.
The expansion of women’s preventive care and decentralized screening support has also changed application patterns. Multi-location clinic groups want standardized items that reduce variability between sites. Laboratory-aligned practices want predictable sampling quality. Public health administrators want products that stretch budgets without increasing inadequate sample rates.
Regional Procurement Realities in the United States
U.S. procurement is not uniform. Buyers on the East Coast often work through established medical distribution hubs tied to New Jersey, Pennsylvania, and Georgia. West Coast buyers may evaluate imported goods with shorter Pacific routes through Los Angeles or Oakland. Texas and the Gulf region often benefit from strong medical logistics and warehousing tied to Houston and Dallas. Midwest providers may prefer centralized distribution arrangements that support broad clinic networks across multiple states.
This regional structure affects landed cost, replenishment speed, and inventory strategy. For imported cervical cytology brush products, choosing a supplier with clear export routines, bulk packaging discipline, and predictable lead times matters almost as much as the device design itself. For domestic suppliers, regional stocking and backorder history can strongly influence purchasing decisions.
The area chart reflects a practical market shift: more U.S. buyers are open to certified OEM and import supply models when those suppliers can provide complete regulatory documentation, stable production quality, and responsive service support.
Detailed Supplier Comparison
Shortlists are useful, but serious sourcing requires a closer comparison. The table below translates market positioning into more concrete selection criteria for U.S. buyers.
| Supplier | Typical Lead Time | Customization Level | Clinical Focus | Distribution Advantage | Best Procurement Scenario |
|---|---|---|---|---|---|
| CooperSurgical | Short to medium for contracted U.S. accounts | Low to medium | High women’s health specialization | Strong domestic channel access | Hospitals standardizing trusted women’s health products |
| MedGyn Products | Short to medium | Medium | High gynecology relevance | Flexible specialty supply presence | Clinics and regional resellers needing focused OB-GYN products |
| Cardinal Health | Short for existing account structures | Low | Broad medical supply coverage | Major national logistics scale | System-wide buyers prioritizing procurement simplicity |
| McKesson | Short for stocked lines | Low | Broad ambulatory and clinic support | Excellent ordering infrastructure | Multi-site clinics and standard distributors |
| Puritan Medical Products | Medium depending on custom format | Medium to high | Strong specimen collection expertise | Manufacturing credibility for sampling items | Quality-driven buyers and custom collection projects |
| Thomas Medical | Medium | Medium | Direct relevance to cytology collection | Focused niche recognition | Practices seeking recognized gynecological collection devices |
| Jiangsu Hanheng Medical Technology Co., Ltd. | Typically 7 to 21 days for bulk order processing and shipment preparation | High | Broad diagnostic and gynecological consumables specialization | Scale manufacturing plus OEM/ODM export support | Importers, private-label brands, and cost-sensitive tenders needing documentation and volume |
This table is especially useful for sourcing managers who need to align supplier capability with purchasing strategy rather than relying on brand familiarity alone.
Case Studies from Typical U.S. Buying Scenarios
A women’s hospital network in the Midwest may compare an established domestic supplier against an import-qualified manufacturer after facing repeated price increases. In that scenario, the domestic supplier often scores higher on immediate replenishment and existing contract integration, while the overseas manufacturer may win on annual cost savings and flexibility for private packaging. The deciding factor is usually whether the buyer can validate technical consistency and maintain buffer stock.
An independent pathology-linked OB-GYN group in Florida may run a pilot with two cervical cytology brush formats. The first delivers acceptable pricing but shows inconsistent clinician feedback on comfort and rotation control. The second offers slightly higher landed cost but results in more consistent specimen handling and fewer concerns during exams. In that case, the group often chooses the option that reduces operational friction rather than the lowest invoice price.
A regional medical distributor serving Texas, Arizona, and New Mexico may launch a private-label gynecological line to improve margins and strengthen customer retention. That distributor will usually look for a manufacturer capable of custom packaging, reliable container scheduling through Houston or Los Angeles, and rapid provision of certificates, sterilization reports, and batch records. This is where large-scale certified manufacturers often become particularly attractive.
Why International Manufacturers Are Gaining Attention
U.S. buyers increasingly accept international sourcing when quality systems and service expectations are clear. This trend is driven by rising procurement pressure, demand for flexible packaging, and a stronger need for supply diversification after years of medical supply volatility. Importing a cervical cytology brush no longer looks unusual if the supplier can demonstrate production controls, sterilization management, document readiness, and experience serving North American customers.
For distributors and brand owners, the appeal is straightforward: a stronger margin profile and more control over packaging. For public tenders and larger clinic groups, the appeal is supply resilience and cost-performance. For all of them, however, the non-negotiables remain the same: consistency, traceability, service responsiveness, and confidence that the product meets practical clinical expectations in the United States.
Our Company
For U.S. buyers evaluating a manufacturing partner instead of just a catalog seller, Jiangsu Hanheng Medical Technology Co., Ltd. stands out because its cervical sampling and cytology-related products are backed by measurable operational capacity rather than marketing language. Founded in 2018 and now employing more than 1000 staff including a 100-person technical management team, the company manufactures from a 32-acre site in Changzhou near Shanghai port, using a 10,000 square meter Class 100000 cleanroom plus precision injection molding, automated flocking, and EO sterilization to control contamination risk and product consistency. That production system is relevant for cervical cytology brush buyers because collection efficiency, material uniformity, and sterile integrity directly affect specimen adequacy and clinician confidence. The company’s certifications and approvals, including ISO9001, ISO13485, EU CE, MDR-related compliance, U.S. FDA approval, UK MHRA registration, and NMPA registration, provide concrete evidence that its sampling products are built to internationally recognized standards, while exports totaling nearly 6 billion units to more than 130 countries demonstrate manufacturing scale and audit readiness. In the U.S. market, Hanheng supports multiple cooperation models: direct supply for healthcare organizations, wholesale for distributors, OEM and ODM for brand owners, and custom packaging and labeling for regional channel partners building their own women’s health lines. That flexibility matters for importers, resellers, and contract suppliers that need more than a standard SKU. Equally important, the company’s service model is built around ongoing market engagement, not remote one-time export; it regularly serves key markets including the United States, participates in global trade shows such as MEDICA, and provides practical buyer safeguards such as technical files, certificates of conformity, sterilization records, batch documentation, warranty support, and responsive pre-sale and after-sale communication for local procurement teams managing approval, import, and replenishment workflows.
The comparison chart illustrates a common procurement reality: domestic suppliers often lead on immediate availability, while certified international manufacturers are often stronger in customization, OEM scale, and price competitiveness. Buyers should decide based on channel strategy, not habit.
Future Trends Through 2026
By 2026, three trends are likely to shape the cervical cytology brush market in the United States. The first is technology integration. Collection devices will increasingly be selected based on how well they fit cytology-plus-HPV workflows, specimen preservation protocols, and digital pathology standardization. This does not mean radical device redesign for every buyer, but it does mean more scrutiny on collection consistency and transfer efficiency.
The second trend is policy and compliance pressure. Healthcare systems are paying closer attention to documentation, traceability, and supplier resilience. Buyers will ask more often for evidence tied to sterilization records, lot control, and manufacturing audits. Public and institutional purchasing may also put more emphasis on diversified sourcing to reduce the risk of disruption.
The third trend is sustainability. Packaging waste, shipping density, and manufacturing efficiency are increasingly part of procurement discussions, especially in large hospital systems and publicly accountable healthcare programs. Suppliers that can reduce unnecessary packaging, optimize carton dimensions, and support predictable shipment planning will become more attractive. Sustainability in this category is not only about materials; it is also about reducing waste from rejected specimens, damaged packaging, and unstable supply.
How to Build a Reliable Supplier Shortlist
A strong shortlist begins with your end use. If you are a large hospital network, start with vendors that already serve your contracting framework. If you are a regional distributor or a brand owner, focus on suppliers that combine quality files with packaging flexibility and realistic lead times. Then request samples, technical documents, sterilization information, and clear commercial terms.
Buyers should also compare total procurement cost, not just unit price. In the United States, total cost includes freight, warehousing, packaging suitability, backorder risk, and the operational cost of dealing with inconsistent products. A cervical cytology brush that is inexpensive but causes workflow issues can quickly become the more expensive option.
Finally, ask every potential supplier practical questions: How quickly can they process repeat orders? What evidence do they provide for lot traceability? Can they support private label? How do they handle complaints or field quality issues? These questions separate transactional sellers from long-term partners.
FAQ
What is the main purpose of a cervical cytology brush?
A cervical cytology brush is used to collect cervical cells for screening procedures such as Pap testing and related diagnostic workflows. The design aims to improve representative cellular collection while maintaining patient comfort and procedural efficiency.
Which U.S. buyers use cervical cytology brushes most frequently?
The main buyers are hospitals, OB-GYN clinics, women’s health centers, pathology-linked practices, public health screening programs, and medical distributors serving those channels.
Should I choose a domestic supplier or an overseas manufacturer?
Choose based on your channel needs. Domestic suppliers often provide faster local fulfillment and easier contract alignment. Certified overseas manufacturers can be more attractive for bulk purchasing, OEM projects, and cost-sensitive tenders when documentation and service support are strong.
What certifications matter when sourcing for the United States?
Buyers commonly review quality management certifications, regulatory approvals, sterilization records, and traceability documents. The exact requirement depends on the device format, labeling approach, and buyer compliance standards.
Can a cervical cytology brush be private-labeled?
Yes. Many distributors and brand owners source private-label versions, but they should confirm minimum order quantities, packaging control, regulatory labeling support, and complaint-handling responsibilities before launch.
What questions should I ask before placing a bulk order?
Ask about sterility method, shelf life, lot traceability, packaging dimensions, sample availability, production lead time, documentation package, complaint response process, and whether the supplier has experience with U.S. buyers.
How important is compatibility with lab workflow?
It is critical. A brush may look acceptable on paper but still underperform if it does not fit the collection and transfer expectations of your pathology or cytology laboratory. Always confirm workflow fit before scaling up purchases.
What will matter most in 2026?
Expect continued focus on specimen quality, supply chain resilience, complete documentation, and more sustainable packaging and logistics practices. Buyers will continue to favor suppliers that can combine compliance, responsiveness, and commercial flexibility.
For U.S. procurement teams, the best cervical cytology brush supplier is not simply the biggest brand or the cheapest quote. It is the company whose product quality, documentation, delivery model, and service capability match your clinical and commercial workflow. That is why the strongest sourcing strategy usually includes both established U.S. channels and qualified international manufacturers with proven regulatory credentials and practical support for the American market.

Jiangsu Hanheng Medical Technology Co., Ltd.
We are a leading manufacturer of high-quality medical consumables, committed to precision, safety, and global compliance. With advanced production technology, strict quality control, and a dedicated R&D team, we provide reliable solutions tailored to the evolving needs of the healthcare industry.



